Chp 5 Sec 4 Guided Notes Rates of Change For a grade Name
advertisement

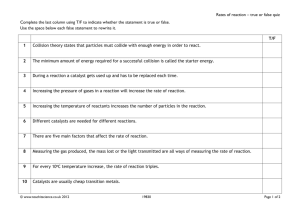
Chp 5 Sec 4 Guided Notes Rates of Change For a grade Name:_____________________________ 1. Chemical reactions can occur at different ________________or rates. 2. Many factors can affect the rates of reactions *6 Factors Affecting Reactions 1. Temperature 2. ____________________ 3. Concentrations 4. Pressure 5. ___________________ 6. Catalysts Temperature: 7. Most reactions go ______________at higher temperatures 8. Example: A potato slice cooks faster in hot oil than in boiling water. Heating food______________ up chemical reactions Increased speed of particles increases chances for particles to_______________& react. Surface Area: 9. A____________________ surface area speeds up reactions 10. An object with a large surface area reacts more rapidly because more particles can come into contact with the other reactants. Concentration: 11. Concentrated solutions react faster 12. Higher concentration ______________________the chance for particles to collide and react. Pressure: 13. Reactions are quicker at _______________________pressure Example: ◦ Gases react________________ at higher pressures, the particles have less space to move and therefore have more collisions. Molecule Size: 1. The size & shape of the reactant molecules affect the rate of reaction. 2. Massive, bulky molecules react____________________ Catalysts: 3. Catalysts can speed up or slow down chemical reactions 4. Catalysts are not reactants or _____________________ 5. Catalysts that slow chemical reactions are called_______________________. Ex. Enzymes Biological _________________ Le Chatelier’s principle predicts changes in_______________________. If a change is made to a system in a chemical equilibrium, the equilibrium shifts to ______________________the change until a new equilibrium is reached. (shifting “left” or “right”) If the temperature is increased, the reaction at equilibrium will shift to the left In the direction that absorbs energy. If the pressure is increased, the reaction will shift to the __________________ and reduce the pressure. Practice Balancing: _____ K + _____ Br2 _____ KBr _____ C7H16 + _____ O2 _____ CO2 + _____ H2O _____ P4 + _____ O2 _____ P2O5 _____ FeS2 + _____ O2 _____ Fe2O3 + _____ SO2 _____ Al + _____ FeO _____ Al2O3 + _____ Fe