1. Atomic Units
advertisement

Atomic units; Molecular Hamiltonian;
Born-Oppenheimer approximation
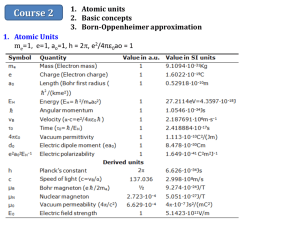
1. Atomic Units
The atomic units have been chosen such that the fundamental electron properties are all
equal to one atomic unit. (me=1, e=1, = h/2π = 1, ao=1, and the potential energy in
the hydrogen atom (e2/4πε0ao = 1).
Symbol
Quantity
Value in a.u.
Value in SI units
me
Mass (Electron mass)
1
9.1094·10-31Kg
e
Charge (Electron charge)
1
1.6022·10-19C
a0
Length (Bohr first radius
1
0.52918·10-10m
2
( /me2))
EH
Energy (EH= 2/mea02)
1
27.2114eV=4.3597·10-18J
Angular momentum
1
1.0546·10-34Js
vB
Velocity (α·c=e2/4πε0 )
1
2.187691·106m·s-1
τ0
Time (τ0= /EH)
1
2.418884·10-17s
4πε0
Vacuum permittivity
1
1.113·10-10C2/(Jm)
d0
Electric dipole moment (ea0)
1
8.478·10-30Cm
e2a02EH-1
Electric polarizability
1
1.649·10-41 C2m2J-1
Derived units
h
Planck’s constant
c
Speed of light (c=vB/α)
μB
Bohr magneton (e /2me)
μN
Nuclear magneton
μ0
Vacuum permeability (4π/c )
E0
Electric field strength
2
2π
6.626·10-34Js
137.036
2.998·108m/s
½
9.274·10-24J/T
2.723·10-4
5.051·10-27J/T
6.629·10
4π·10-7 Js2/(mC2)
-4
1
5.1423·1011V/m
In atomic units the mass of a proton (1.6726 x 10-27 kg) would be 1836.15 au, and the
reduced mass of the hydrogen molecule (.503913 g/mol) would be 925.260 au.
The use of atomic units also simplifies Schrödinger's equation. For example the
Hamiltonian for an electron in the Hydrogen atom would be:
SI units:
2
1 e2
2
2me
4 0 r
Atomic units:
1 2 1
2
r
Other frequently used energy units:
1a.u. = 27.212eV = 627.51Kcal/mol = 219470 cm-1
1Kcal/mol = 4.184KJ/mol
Debye: 1ea0 = 2.54181De; 1De=0.3934ea0
Other fundamental constants:
Boltzmann’s constant:
k=1.38066·10-23J/K
Avogadro’s number:
NA=6.02205·1023mol-1
Rydberg constant:
R∞=1.097373·107m-1
Compton wavelength of electron:
λC=2.426309·10-12m
Stefan-Boltzmann constant: σ=5.67032·108W/(m2K4)
1
Electric field unit:
Field=X+a means that an electric field of a*10-4 a.u. is applied along the X direction.
1.a.u.=Hartree/(charge*bohr) = 27.2114*1.6 10-19J/(1.6 10-19C * 5.29177 10-11 m)
1 a.u. = 5.1423 1011 V/m
1 a.u. = 51.423 V/Å
Ef(V/Å)=51.423Ef(a.u.)
Field=X+1000 means a field of 0.1a.u. (5.1423V/Å) is applied along the X direction
2. Basic concepts
- structure of many-electron operators (e.g. Hamiltonian)
- form of many-electron wave-functions
(Slater determinants, and linear combination of them)
- Hartree-Fock (HF) approximation
- more sophisticated approaches which use the HF method as a starting point
(correlated post-Hartree Fock methods)
Aproximations made in the framework of the Hartree-Fock-Roothaan-Hall theory
2
Molecular Hamiltonian
The non-relativistic time-independent Schrödinger equation:
H|Ψ>=E|Ψ>
H – Hamiltonian operator for a system of nuclei and electrons
riA | riA || ri RA |
rij | rij || ri r j |
R AB | R AB || R A RB |
i, j – electrons (N)
A, B – nuclei (M)
A molecular coordinate system
N
M
N
M
N 1 N
M 1 M
ZA
Z A ZB
1 2
1
1
H
i
2A
2
2M
riA
r
R B
i 1
A
1 A
i 1
A
1
i 1 j i ij
1 B
A A
A
Te
2
2
2
i2 2
x
y i2
zi2
i
TN
VeN
Vee
(1)
VNN
MA - the ratio of the mass of nucleus A to the mass of an electron
ZA – the atomic number of nucleus A
Te – the operator for the kinetic energy of the electrons
TN – the operator for the kinetic energy of the nuclei
VeN – the operator for the Coulomb attraction between electrons and nuclei
Vee – the operator for the repulsion between electrons
VNN – the operator for the repulsion between nuclei
(1)– represents the general problem
to be separated in two parts: electronic and nuclear problems
3
Born-Oppenheimer Approximation
Ψ=Ψ(x1,…,xN, X1,…,XM)
The term VeN in the Hamiltonian prevents any wave-function Ψ(x,X) solution of the time
independent Schrödinger equation from being written as a product of an electronic
wavefunction and a nuclear wavefunction.
Thus, we need approximations!
The nuclei are much heavier than electrons (mproton=1836me)
they move much more slowly
the nuclei can be considered frozen in a single arrangement
(molecular conformation)
the electrons can respond almost instantaneously to any change
in the nuclear position
The electrons in a molecule are moving in the field of fixed nuclei. Neglecting the spin of
nuclei, the total wavefunction is written as:
Ψ=Ψe(x,R)ΨN(R)
On this basis:
► 2-nd term in (1) can be neglected
► 5-th term in (1) is a constant
Electronic Hamiltonian:
-
describes the motion of N electrons in the field of M fixed point charges
He (R )
N
i 1
1 2
i
2
N
M
Z A N 1
r
A 1 iA
i 1
i 1
N
r
1
ji
(2)
ij
He(R) means that He depends on the nuclei positions
Electronic Schrödinger equation:
He(R)Ψe(x;R)=Ee(R)Ψe(x;R)
(3)
Ψe=Ψe(x;R)
(4)
Ee = Ee(R)
(5)
(4) - is the electronic wave-function which describes the motion of the electrons
- explicitly depends on the electronic coordinates
- parametrically depends on the nuclear coordinates because H is a function of
the positions R of the nuclei
Another approximation: for each value of the nuclear positions, the electronic system is
in the electronic ground state, corresponding to the lowest Ee(R).
parametric dependence
-
the nuclear coordinates do not appear explicitly in Ψe.
-
from the point of view of the electrons, the nuclear degrees of freedom are fixed
-
a different electronic wave-function is obtained for each nuclear configuration
4
The total energy:
Epot
tot (R ) E e
M 1 M
Z A ZB
RAB
A 1 B A
(6)
Equations (2) – (6) ≡ electronic problem
This geometry dependent energy is generally termed potential energy surface (PES)
and represents the effective potential that determines the nuclear motion.
If the electronic problem is solved
► we can solve for the motion of the nuclei using the electronic energy E(R) as
the potential energy in Schrödinger equation for the nuclear motion.
Since the electrons move much faster than the nuclei
► we can replace the electronic coordinates by their average values (averaged
over the electronic wave-function)
nuclear Hamiltonian
describes the motion of the nuclei in the average field of the electrons
o
M
HN
2M
N
1
A 1
A
2A
1
i 1
M
1
2A E e ({R})
2
M
A
A 1
M
2M
A 1
1
A
N
M
ZA
2 r
2
i
i 1 A 1 iA
N 1 N
r
i 1 j i
1
ij
M 1 M
Z A ZB
RAB
A 1 B A
M 1 M
Z A ZB
RAB
A 1 B A
2A Epot
tot ({R})
Potential energy surface (PES)
Etotpot ({R})
Schematic illustration of a potential energy surface
The equilibrium conformation of the molecule corresponds to the minimum of the
potential energy curve
5
nuclear Schrödinger equation
HN|ΨN> = E|ΨN>
ΨN - describes the vibration, rotation and translation of a molecule
E
- total energy of the molecule (in the Born-Oppenheimer approximation)
- includes:
- electronic energy
- vibrational energy
- rotational energy
- translational energy
Total wave-function in the Born-Oppenheimer approximation:
Ψ(x,R) = Ψe(x,{R})·ΨN(R)
Born-Oppenheimer approximation
-
usually a good approximation
-
bad approximation for:
o
excited states
o
degenerate or cuasidegenerate states
Requirements for the wave function
We will assume the Born-Oppenheimer approximation and will only be concerned with
the electronic Schrödinger equation.
1. Normalization
*
(r )(r )dr 1
Integration is performed over the coordinates of all N electrons.
The wavefunction must also be single-valued, continuous and finite.
2. Antisymmetry with respect to the permutation of two electrons
Electrons are fermions and a many electron wave-function must be antisymmetric with
respect to the interchange of the coordinate x (both space and spin) of any two
electrons.
Ψ(x1, x2, ... , xi, ..., xj, ...,xN) = -Ψ(x1, x2, ... , xj, ..., xi, ...,xN)
The Pauli exclusion principle is a direct result of this antisymmetry principle.
3. The electronic wavefunctions must be eigenfunctions of Sz and S2 operators,
with the eigenvalues MS and S(S+1).
Since the electronic Hamiltonian does not contain any spin operators, it does commute
with the operators Sz and S2 defined below, with the corresponding eigenvalues MS and
S(S+1), respectively.
6
N
Sz
s
i
N
zi
MS
S2
s
2
i
S(S 1)
i
[H,Sz]=0
[H,S2]=0
α(σ) and β(σ) – spin functions (complete and orthonormal)
( ) ( )d ( ) ( )d 1
1
and
( ) ( )d ( ) ( )d 0
0
The electron is described by spatial (r) and spin (σ) coordinates:
x={r,σ}
Homework
1. Write the Z-matrix for the tetramethylsilane
(TMS=Si(CH3)4) molecule in Td symmetry.
2. Use the Gaussian program and obtain the PES of the water
molecule in the C2v symmetry by varying the bond length from 0.9 to 1.1 and the bond
angle from 100o to 110o. Plot the PES using a dedicated software (Mathematica,
MathLab, Mathcad).
3.
Write
a
C
program
to
calculate
the
coordinates of the atoms in a graphene sheet
(10x10 atoms) whose structure is given below.
The C-C bond length in graphene is 1.42Å.
7