doc - King Fahd University of Petroleum and Minerals
advertisement

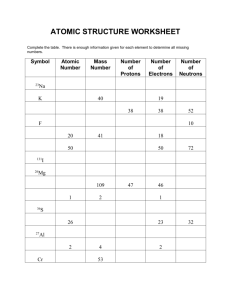
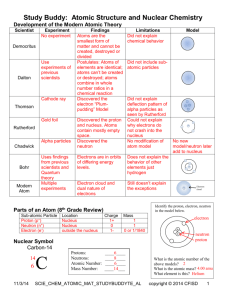
KING FAHD UNIVERSITY OF PETROLEUM AND MINERALS DEPARTMENT OF PHYSICS PHYS 353-062 HOMEWORK No. 1 DUE TUESDAY 27 FEB 2007 Show the details of the your calculations. 1. What is the closest approach that a 5.3 MeV alpha particle can make to a gold nucleus? 2. Calculate the number of atoms per cubic centimeter of lead given that the density of lead is 11.3 g/cm3 and its atomic mass is 207.21 amu. 3. What is the uncertainty in the momentum of a proton inside a nucleus of 27Al? What is the kinetic energy of this proton? 4. The density of beryllium (Z = 4) is 1.84 g/cm3, and the density of lead (Z = 82) is 11.3 g/cm3. Calculate the nuclear density of a 9Be and 208Pb nucleus. 5. Determine the electronic shell configuration for aluminum (Z = 13). 6. What is the difference in mass between the hydrogen atom and the sum of the masses of a proton and an electron? Express the answer in energy eV. 7. The atomic mass of tritium is 3.017005 amu. How much energy in MeV is required to dissociate the tritium into its component parts? 8. The atomic masses of 16O, 17O, and 18O are 15.994915, 16.999131, and 17.999160 amu, respectively. Calculate the atomic mass of the element oxygen. 9. Calculate the molecular mass of chlorine, Cl2, using the exact atomic masses of the chlorine isotopes given in appropriate reference sources. 10. What is the binding energy of the last neutron in 17O?