Metric Measures
advertisement

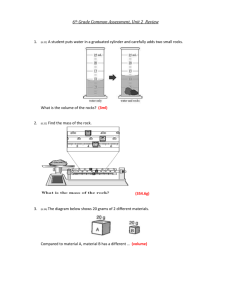
Metric Measures Length, Mass (Weight) & Density PART I MASS Mass of an object, the amount of material it is, is closely related to its weight (a relation of mass and gravity). Weighing an object with a scale or balance is finding its mass "here on earth". Weights today will be measured using a triple beam balance unless otherwise requested. Weights should be recorded to the nearest 0.01g (gram) on this scale. A. Lead Object a. Obtain a lead object from the instructor. Keep this item; you will be making further measurements on it in Part II. b. Obtain a weighboat. Measure and record its mass. c. Place your lead object in the weighboat. Measure and record this new (combined) mass. d. Find, by subtraction, the mass of the lead object, record it. B. Analytical Balance use (card & thumbprint, monofilament): a. Obtain a "card" or piece of paper provided. Do NOT touch it; use tweezers to move it. b. Using the analytical balance, measure and record the card’s mass to the nearest 0.0001g. c. Move the card off the scale. d. Place your thumb print on the card and apply fingerprint powder. Remove excess powder. e. Print your full name on the card (use a pencil or ballpoint pen). f. Reweigh your card. Record this new (combined) mass. g. Staple your thumbprint card to the back of your lab data sheet. h. Find, by subtraction, the mass of your (processed) fingerprint and signature, record it. i. Find and record the mass of the three different strands of monofilament provided. Return each to its proper envelope when finished. C. Electronic Balance use: a. Use a weighboat for all measurements. Tare (zero) out the weighboat before placing objects into it for mass measurements. b. Measure and record the mass of Object A in grams and in ounces (toggle the readout mode!). c. Measure and record the mass of Object B in grams and in ounces (toggle the readout mode!). d. Measure and record the mass of Object C in grams and in ounces (toggle the readout mode!). PART II DENSITY Density of an object is defined as its mass per unit volume. D=M/V The mass is measured in grams (g) and the volume is measured in cubic centimeters (cm3 or cc) giving the units of g/cm3. Density of Liquids A. Water: a. Weigh a clean, dry 10mL graduated cylinder. Record this weight. b. Add tap water to your cylinder until you have (approximately) 8mL. Read and record the exact volume of water in the cylinder to the nearest 0.1mL. [Read the bottom of the meniscus or downwardly curved surface. Have the surface at eye level or eye at surface level!] c. Find and record the mass of the cylinder and water. d. Find, by subtraction, the mass of the water, record it. e. Compute and record the density of the sample of water. B. Diesel Fuel or Mineral Spirits or Kerosene (as assigned): a. Repeat the above procedures using about 8mL of the assigned fluid. Density of Solids A. Wood Block a. Obtain a wood block and metric rule. Record the Block’s number. Measure and record its length (L), width (W), and height (H) to the nearest 0.1cm. b. Compute and record the volume of the Block. (vol = L x W x H) c. Measure and record the mass of the Block. d. Calculate and record the density of the block. B. Lead Object a. To a clean dry 10mL graduated cylinder add about 2-3mL of water. Read and record the volume of water to the nearest 0.1mL. b. Carefully place your lead object into the cylinder of water being careful in not splashing any water out or up onto the cylinder sides. c. Read and record this new volume. d. Find, by subtraction, the volume of the lead object [amount of water displaced]. [This is the water displacement technique for finding the volume of an irregularly shaped object.] e. Calculate and record the object’s density. Remember that you have already measured the object’s mass in Part I. f. Obtain and record the density of lead from the Handbook of Chemistry and Physics. g. Compare the two values for the density of lead (e. and f.). Calculate and record the raw difference and the % difference between these two values. % difference = (100 x [theoretical value – experimental value]) / theoretical value Name________________________ Section________________ Metric Measures Length, Mass (Weight) & Density PART I OBJECT MASSING A. Lead Object Mass of weightboat + lead object _________________ Mass of weighboat _________________ Mass of lead object _________________ B. Card & thumbprint & signature [analytical scale] Mass of card + thumbprint + signature _________________ Mass of card _________________ Mass of thumbprint + signature _________________ Mass of monofilament A _________________ Mass of monofilament B _________________ Mass of monofilament C _________________ C. Electronic top loading scale Grams Ounces Mass of Object A _________________ _________________ Mass of Object B _________________ _________________ Mass of Object C _________________ _________________ Name________________________ PART II Section________________ DENSITY A. Liquids Water Liquid #2 (________________) Mass of graduated cylinder + liquid _________________ _________________ Mass of graduated cylinder _________________ _________________ Mass of liquid _________________ _________________ Volume of liquid _________________ _________________ Density of liquid _________________ _________________ length: ________________ width: ________________ height: ________________ mass: ________________ volume: ________________ density: ________________ B. Wood Block # ____________ C. Lead Object Volume before immersion _________________ Volume after immersion _________________ Mass of lead object _________________ Volume of lead object _________________ Density of lead object _________________ Density of lead (handbook value) _________________ Difference of densities _________________ % difference of densities ________________