Chemicals & Chemical Change: Chemistry Basics Presentation
advertisement

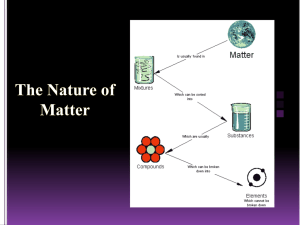
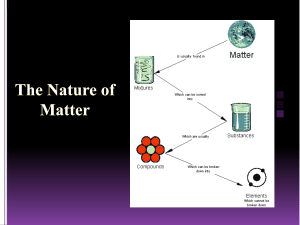
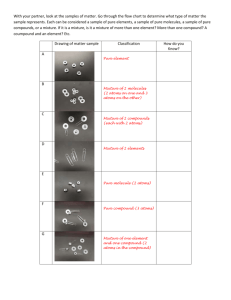
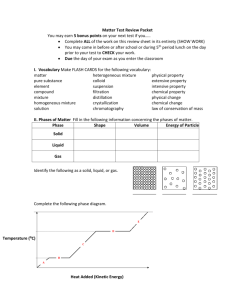
Chemicals & Chemical Change Chemistry? • Chemistry is the study of matter. • Matter is anything that has mass and takes up space (volume). • All matter is made up of tiny particles called atoms. (Atoms must have a mass and take up space.) Classifying Matter Classifying Matter • Pure Substance – A substance in which all of the particles are the same. – Element – Pure substance that is made of only one type of atom and it can’t be broken down into a simpler substance. – Compound – Pure substance that contains two or more different elements in a fixed ratio/proportion. • Mixture – A substance that is made of two or more different types of particles. – Homogeneous mixture (AKA Solution) – A mixture that looks the same throughout – it has only one phase. – Heterogeneous mixture – A mixture that has a non-uniform composition. The different parts are easily identifiable and can usually be separated. Properties of Matter • Properties are characteristics that can be used to describe something. • A physical property is a characteristic of a substance that can be described when the substance is by itself. – State, colour, hardness, odour, melting and boiling point are some physical properties. • A chemical property is a characteristic behaviour that occurs when the substance is exposed to another substance. – Combustibility, reactivity with acids, corrosive and the ability to oxidize are chemical properties. Chemical vs. Physical Change • A physical change occurs when a substance undergoes a change in size or state. The substance does not change into another substance – it stays the same! H2O (l) H2O (s) • A chemical change occurs when a substance undergoes a change and becomes a new substance with new properties. 2Na (s) + Cl2 (g) 2NaCl (s) Chemical Tests • In your notes… – Indicate what a chemical test is used for in the lab. – Outline the chemical tests for oxygen, hydrogen, carbon dioxide and water. Lab Safety Symbols • • • • • • • • • Poison Flammable Explosive Corrosive Toxic Highly Reactive Oxidizing Agent Biohazard Radioactive That’s All I Got…