Practice Problem - HCC Southeast Commons
advertisement

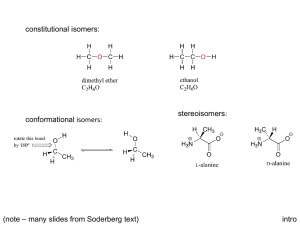
Chapter 9 Introduction • Isomers are compounds with the same chemical formula but different structures Constitutional isomers • Constitutional isomers are isomers with different order of connections: skeletal, functional and positional Stereoisomers • Stereoisomers are isomers with same connections, but different spatial arrangement of atoms – Enantiomers – are nonsuperimposable mirror images – Diastereomers – are all other stereoisomers – include cis, trans and configurational Introduction to Chirality • Some objects are not the same as their mirror images (technically, they have no plane of symmetry) – A right-hand glove is different than a left-hand glove – The property is commonly called “handedness” • Organic molecules (including many drugs) have handedness that results from substitution patterns on sp3 hybridized carbon • Molecules exist as three-dimensional objects – Some molecules are the same as their mirror image – Some molecules are different than their mirror image These are stereoisomers called enantiomers A. The Tetrahedral Carbon • Enantiomers - are molecules that are not the same as their mirror image – They are the “same” if the positions of the atoms can coincide on a one-to-one basis • We test if they are superimposable, which is imaginary • This is illustrated by enantiomers of lactic acid Mirror-image forms of Lactic Acid • When H and OH substituents match up, COOH and CH3 don’t • When COOH and CH3 coincide, H and OH don’t • Molecules that have one carbon with 4 different substituents have a nonsuperimposable mirror image – enantiomer • Build molecular models to see this B. Chirality • Chiral molecules - are not superimposable with their mirror images They have handedness They lack a plane of symmetry A molecule is not chiral if it has a plane of symmetry • A plane of symmetry divides an entire molecule into two pieces that are exact mirror images – If an object has a plane of symmetry it is necessarily the same as its mirror image – An achiral molecule is a molecule with a plane of symmetry and is the same as its mirror image • The plane has the same thing on both sides for the flask • There is no mirror plane for a hand Chirality • Chirality - is the lack of a plane of symmetry - is also called “handedness” - Hands, gloves are prime examples of chiral objects They have a “left” and a “right” version Chirality Centers • Chirality center - is a point in a molecule where four different groups (or atoms) are attached to carbon - is also called “asymmetric center”, “stereogenic center”, or “stereocenter” • There are two nonsuperimposable ways that 4 different groups (or atoms) can be attached to one carbon atom – If two groups are the same, then there is only one way Chirality Centers in Chiral Molecules • A chiral molecule usually has at least one chirality center • Groups are considered “different” if there is any structural variation • In cyclic molecules, we compare by following in each direction in a ring Practice Problem: Identify the chirality centers in the following molecules. Practice Problem: Alanine, an amino acid found in proteins, is chiral. Draw the two enantiomers of alanine using the standard convention of solid, wedged, and dashed lines. Practice Problem: Identify the chirality centers in the following molecules (red = O, yellow-green = Cl, pale yellow = F) C. Optical Activity • Optical Activity - is the ability of a molecule to rotate the plane of a polarized light Light restricted to pass through a plane is planepolarized Phenomenon discovered by Biot in the early 19th century • In the 19th century, Biot observed that: – Achiral compounds do not change the plane of the plane-polarized light and they are said to be optically inactive – Chiral compounds rotate the plane of the planepolarized light and they are said to be optically active Optical Activity • It is measured with a polarimeter – Light passes through a plane polarizer – Plane polarized light is rotated in solutions of optically active compounds • Rotation, in degrees, is [] – Clockwise rotation is called dextrorotatory (+) – Counterclockwise rotation is levorotatory (-) Measurement of Optical Rotation • A polarimeter measures the rotation of planepolarized light that has passed through a solution – The source passes through a polarizer and then is detected at a second polarizer (analyzer) – The angle between the entrance and exit planes is the optical rotation. A Simple Polarimeter • It measures extent of rotation of plane polarized light • Operator lines up polarizing analyzer and measures angle between incoming and outgoing light Specific Rotation • To have a basis for comparison, we use specific rotation, []D for an optically active compound: Observed rotation, (degrees) []D = ______________________________ Pathlength, l (dm) x Concentration, C (g/ml) ( o) ____________ []D = l (dm) x C (g/ml) Specific Rotation • Specific rotation, []D for an optically active compound is that observed for: – C = 1 g/mL in solution in cell – l = 10 cm path l = 589 nm (yellow light emitted from sodium metal vapor) Specific Rotation • For a compound to be optically active, the compound must be chiral Specific Rotation • The specific rotation of the enantiomer is equal in magnitude but opposite in sign []D = +3.82o []D = -3.82o Practice Problem: A 1.50 g sample of coniine, the toxic extract of poison hemlock, was dissolved in 10.0 mL of ethanol and placed in a sample cell with 5.00 cm pathlength. The observed rotation at the sodium D line was +1.21o. Calculate []D for coniine. D. Pasteur’s Discovery of Enantiomers • 1849: Louis Pasteur discovered that sodium ammonium salts of tartaric acid crystallize into right handed and left handed forms – The optical rotations of equal concentrations of these forms are opposite • The solutions contain mirror image isomers, called enantiomers and they crystallized in distinctly different shapes • Enantiomers are physically identical in all respects except for their effect on plane-polarized light. – They have the same melting point, the same boiling point, the same solubilities, and the same spectroscopic properties. • A 50:50 mixture of two enantiomers is optically inactive Relative 3-Dimensional Structure • The original method was a correlation system, classifying related molecules into “families” focused on carbohydrates – Correlate to D- and Lglyceraldehyde – D-erythrose is the mirror image of L-erythrose • This does not apply in general E. Sequence Rules for Specification of Configuration • When naming a molecule, the three-dimensional arrangement of atoms or configuration at a chirality center is also needed. – Cahn-Ingold-Prelog sequence rule is used for the specification Steps in determining a configuration at a chiral atom1 • Rule 1: Look at the four atoms directly attached to the chirality center, and assign priorities in order of decreasing atomic number (or mass number). Steps in determining a configuration at a chiral atom2 • Rule 2: If a decision about priority cannot be reached by applying rule 1, compare atomic numbers of the 2nd atoms in each substituent, continuing on as necessary until the 1st point of difference. Steps in determining a configuration at a chiral atom3 • Rule 3: Multiple-bonded atoms are equivalent to the same number of single-bonded atoms. Sequence Rules (IUPAC) • Assign each group priority according to the CahnIngold-Prelog scheme with the lowest priority group pointing away, look at remaining 3 groups in a plane • Clockwise is designated R (from Latin for “right”) • Counterclockwise is designated S (from Latin word for “left”) The sign of optical rotation, (+) or (-), is not related to the R, S designation Practice Problem: Assign priorities to the following sets of substituents: a. -H, -Br, -CH2CH3, -CH2CH2OH b. -CO2H, -CO2CH3, -CH2OH, -OH c. -CN, -CH2NH2, -CH2NHCH3, -NH2 d. -Br, -CH2Br, -Cl, -CH2Cl Practice Problem: Orient each of the following drawings so that the lowest-priority group is toward the rear, and then assign R or S configuration: Practice Problem: Assign R or S configuration to the chirality center in each of the following molecules: Practice Problem: Draw a tetrahedral representation of (S)-2pentanol (2-hydroxypentane). Practice Problem: Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (red = O, blue = N, yellow = S): F. Diastereomers • Diastereomers are stereoisomers that are not mirror images of each other Can you identify the diastereomers? • Molecules with more than one chirality center have mirror image stereoisomers that are enantiomers 2R,3R • In addition they can have stereoisomeric forms that are not mirror images, called diastereomers 2R,3S 2S,3S 2S,3R Practice Problem: Assign R,S configurations to each chirality center in the following molecules. Which are enantiomers, and which are diastereomers? Practice Problem: Chloramphenicol, a powerful antibiotic isolated in 1949 from the Streptomyces venezuelae bacterium, is active against a broad spectrum of bacterial infections and is particularly valuable against typhoid fever. Assign R,S configurations to the chirality centers in chloramphenicol. Practice Problem: Assign R,S configuration to each chirality center in the following molecular model of the amino acid isoleucine (red = O, blue = N) G. Meso Compounds • Meso compounds are compounds that are achiral, yet contain chirality centers – They have a plane of symmetry 2n - 1 • Tartaric acid has two chirality centers and two diastereomeric forms Chiral Achiral • The two structures on the right in the figure are identical so the compound (2R, 3S) is achiral Practice Problem: Which of the following structures represent meso compounds? Practice Problem: Which of the following have a meso form? a. 2,3-Dibromobutane b. 2,3-Dibromopentane c. 2,4-Dibromopentane Practice Problem: Does the following structure represent a meso compound? If so, indicate the symmetry plane (red = O) H. Molecules with More than Two Chirality Centers • A molecule with n chirality centers has a maximum of: – 2n stereoisomers, or – 2n-1 pairs of enantiomers • Cholesterol has 28 = 256 stereoisomers – Only one is naturally important • Molecules can have very many chirality centers • Each point has two possible permanent arrangements (R or S), generating two possible stereoisomers Practice Problem: How many chirality centers does morphine have? How many stereoisomers of morphine are possible in principle? I. Physical Properties of Stereoisomers • Enantiomers differ in the direction in which they rotate plane polarized light but their other common physical properties are the same (+ and -) • Diastereomers have a complete set of different common physical properties (+ and meso/ - and meso) J. Racemic Mixtures and their Resolution • A racemic mixture is a 50:50 mixture of two chiral compounds that are mirror images – It does not rotate light (+) – It is named for “racemic acid” that was the double salt of (+) and (-) tartaric acid • Resolution is the process by which a racemic mixture is separated into its two pure enantiomers To separate components of a racemate (reversibly) 1. We make a derivative of each with a chiral substance that is free of its enantiomer (resolving agent) 2. This gives diastereomers and these are separated by their differing chemical and physical properties. 3. The resolving agent is then removed Acid + Base (Achiral) Salt (Enantiomers) Acid + Base (Chiral) Salt (Diastereomers) Practice Problem: What stereoisomers would result from reaction of (+)-lactic acid with (S)-1-phenyl ethylamine, and what is the relationship between them? Practice Problem: What kinds of isomers are the following pairs? a. (S)-5-Chloro-2-hexene and chlorocyclohexane b. (2R,3R)-Dibromopentane and (2S,3R)-dibromopentane K. Stereochemistry of Reactions • Many reactions can produce new chirality centers from compounds without them – What is the stereochemistry of the chiral product? – What relative amounts of stereoisomers form? Addition of HBr to Alkenes Achiral Intermediate Gives Racemic Product • Addition is via carbocation (sp2-hybridized and planar) • Top and bottom are equally accessible The two transition states are mirror images • Transition states are mirror images and product is racemic Addition of Br2 to Alkenes • Bromine adds to alkenes to give 1,2-dihalides – via three-membered ring bromonium ion intermediate – anti stereochemistry cis-2-Butene: Bromonium Intermediate Gives a Racemic Mixture • Stereospecific • Bromonium ion leads to trans addition trans-2-Butene: Bromonium Intermediate Gives a Meso product • Gives meso product (both are the same) (achiral) • Optically inactive reactants give optically inactive products: – Racemic mixture – Meso product What is the relationship between the products? Practice Problem: Addition of Br2 to an unsymmetrical alkene such as cis-2-hexene leads to racemic 2,3dibromohexane, even though reaction of Brion with the unsymmetrical bromonium ion intermediate is not equally likely at both ends. Make drawings of the intermediate and the products, and explain the observed stereochemical result. Practice Problem: Predict the stereochemical outcome of the reaction of Br2 with trans-2-hexene, and explain your reasoning. Addition of HBr to a Chiral Alkene • Reaction of a chiral reactant with an achiral reactant leads to unequal amounts of diastereomeric products (optically active mixture) Reaction of a chiral reactant with an achiral reactant leads to unequal amounts of diastereomeric products. • Facial approaches are different in energy – One has more steric strain Practice Problem: What products are formed from the reaction of HBr with racemic (+)-4-methyl-1-hexene? What can you say about the relative amounts of the products? Is the product mixture optically active? Practice Problem: What products are formed from reaction of HBr with 4-methylcyclopentene? What can you say about the relative amounts of the products? L. Chirality at Atoms: Other Than Carbons • Trivalent nitrogen – It is tetrahedral – It does not form a chirality center since it rapidly flips • Trivalent Phosphorus – It also applies to phosphorus but it flips more slowly M. Chirality in Nature • Enantiomers have similar physical properties but different biological properties. • Properties of drugs depend on stereochemistry Trade name: Prozac • Stereoisomers are readily distinguished by chiral receptors in nature N. Prochirality • A prochiral molecule - is a molecule that is achiral but that can become chiral by a single alteration Prochiral distinctions: faces • Planar faces that can become tetrahedral are different from the top or bottom • A center at the planar face at a carbon atom is designated re if the three groups in priority sequence are clockwise, si if they are counterclockwise Prochiral distinctions, paired atoms or groups • A prochirality center - is an sp3 carbon with two groups that are the same • To distinguish between the identical groups on a prochirality center, imagine an increase in priority in comparison with the other – If the center becomes R, the group is pro-R – If the center becomes S, the group is pro-S Prochiral distinctions in nature • Biological reactions often involve making distinctions between prochiral faces or groups • Chiral entities (such as enzymes) can always make such a distinction • Example: Addition of H2O to fumarate gives malate (OH on the si face) • Example: Oxidation of ethanol to acetaldehyde (removal of pro-R H) Practice Problem: Identify the indicated hydrogens in the following molecules as pro-R or pro-S: Practice Problem: Identify the indicated faces in the following molecules as re or si: Practice Problem: Lactic acid buildup in tired muscles results from reduction of pyruvate. If the reaction occurs from the re face, what is the stereochemistry of the product? Chapter 9