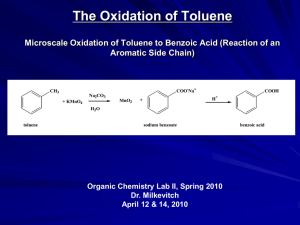
aromatic - HCC Southeast Commons
advertisement

Chapter 15 Introduction • In early 19th century, the term aromatic was used to describe some fragrant compounds – Not correct: later they are grouped by chemical behavior (unsaturated compounds that undergo substitution rather than addition). coal distillate cherries, peaches and almonds Tolu balsam • Currently, the term aromatic is used to refer to the class of compounds related structurally to benzene – They are distinguished from aliphatic compounds by electronic configuration steroidal hormone analgesic tranquilizer 1. • Sources of Aromatic Hydrocarbons There are two main sources of simple aromatic hydrocarbons: i. coal ii. petroleum i. High temperature distillation of coal tar – Coal is a mixture of benzene-like rings joined together. Under high temperature, it produces coal tar which, upon fractional distillation, yields: ii. Heating petroleum at high temperature under high pressure over a catalyst – Petroleum consists mainly of alkanes which, at high temperature under pressure over a catalyst, convert into aromatic compounds. 2. Naming Aromatic Compounds • Aromatic compounds are named according to the system devised by the International Union of Pure and Applied Chemistry (IUPAC). • Aromatic compounds have many common names that have been accepted by IUPAC: • Toluene = methylbenzene • Phenol = hydroxybenzene • Aniline = aminobenzene Monosubstituted benzenes • Monosubstituted benzenes, like hydrocarbons, are systematically named with –benzene as the parent name C6H5Br C6H5NO2 C6H5CH2CH2CH3 Arenes • Arenes are alkyl-substituted benzenes – If # Csubstituent < or = 6, then the arene is named as an alkyl-substituted benzene – If # Csubstituent > 6, then the arene is named as a phenyl-substituted alkane Aryl groups • “Phenyl” refers to C6H5 – It is used when a benzene ring is a substituent – “Ph” or “f” can also be in place of “C6H5 ” • “Benzyl” refers to “C6H5CH2 ” Disubstituted benzenes • Relative positions on a disubstituted benzene ring: – ortho- (o) on adjacent carbons (1,2 disubstituted) – meta- (m) separated by one carbon (1,3 disubstituted) – para- (p) separated by two carbons (1,4 disubstituted) • The ortho- (o), meta- (m), and para- (p) nomenclature is useful to describe reaction patterns Example: “Reaction of toluene with Br2 occurs at the para position” Multisubstituted benzenes • Multisubstituted benzenes (more than two substituents) are named as follows: – Choose the sequence when the substituents have the lowest possible number – List substituents alphabetically with hyphenated numbers – Use common names, such as “toluene”, as parent name (as in TNT) – Use common names, such as “toluene”, as parent name The principal substituent is assumed to be on C1 See Table 15.1 Practice Problem: Tell whether the following compounds are ortho-, meta-, or para-disubstituted (a) Meta (b) Para (c) Ortho Practice Problem: Give IUPAC names for the following compounds (a) m-Bromochlorobenzene (b) (3-Methylbutyl)benzene (c) p-Bromoaniline (d) 2,5-Dichlorotoluene (e) 1-Ethyl-2,4-dinitrobenzene (f) 1,2,3,5-Tetramethylbenzene Practice Problem: Draw structures corresponding to the following IUPAC names: a) p-Bromochlorobenzene b) p-Bromotoluene c) m-Chloroaniline d) 1-Chloro-3,5-dimethylbenzene 3. Structure and Stability of Benzene • Benzene is very stable – It undergoes substitution rather than the rapid addition reaction common to compounds with C=C, suggesting that in benzene there is a higher barrier – Example: Benzene reacts slowly with Br2 to give bromobenzene (where Br replaces H) Heats of Hydrogenation as Indicators of Stability • The addition of H2 to C=C normally gives off about 118 kJ/mol – 3 double bonds would give off 356 kJ/mol – Two conjugated double bonds in cyclohexadiene add 2 H2 • Benzene has 3 unsaturations but gives off only 206 kJ/mol on reacting with 3 H2 molecules – Therefore it has about 150 kJ/mol more “stability” than an isolated set of three double bonds • Benzene has 150 kJ/mol more “stability” than expected for “cyclohexatriene” Benzene’s Unusual Structure • All its C-C bonds are the same length: 139 pm — between single (154 pm) and double (134 pm) bonds • Electron density in all six C-C bonds is identical • Structure is planar, hexagonal • All C–C–C bond angles are 120° • Each C is sp2-hybridized and has a p orbital perpendicular to the plane of the six-membered ring Drawing Benzene and Its Derivatives • The two benzene resonance forms can be represented by a single structure with a circle in the center to indicate the equivalence of the carbon–carbon bonds – This does not indicate the number of electrons in the ring but shows the delocalized structure – One of the resonance structures will be used to represent benzene for ease in keeping track of bonding changes in reactions 4. Molecular Orbital Description of Benzene • In benzene, 6 p orbitals combine to form 6 molecular orbitals (MO): – 3 bonding orbitals with 6 electrons – 3 antibonding orbitals – 3 bonding, low-energy MO: y1, y2, and y3 – 3 antibonding high-energy MO: y4*, y5*, and y6* • Orbitals with the same energy are degenerate – 2 bonding orbitals, y2 and y3 – 2 antibonding orbitals, y4* and y5* – y3 and y4* have no electron density on 2 carbons because of a node passing through these atoms Practice Problem: Pyridine is flat, hexagonal with bond angles of 120°. It undergoes electrophilic substitution rather than addition and generally behaves like benzene. Draw the orbitals of pyridine. Recall: Key Ideas on Benzene • Benzene is a cyclic conjugated molecule • Benzene is unusually stable - DHhydrogenation = 150 kJ/mol less negative than a cyclic triene • Benzene is planar hexagon: bond angles are 120°; carbon–carbon bond lengths, 139 pm • Benzene undergoes substitution rather than electrophilic addition • Benzene is a resonance hybrid with structure between two line-bond structures • Benzene has 6 electrons, delocalized over the ring 5. Aromaticity and the Hückel 4n + 2 Rule The Hückel 4n + 2 rule: – was devised by Eric Hückel in 1931 – states that planar, monocyclic conjugated systems with a total of 4n + 2 electrons where n is an integer (n = 0, 1, 2, 3,…) are aromatic Aromatic compounds with 4n + 2 electrons • Benzene – It has 6 electrons: 4n + 2 = 6, thus n = 1 – It is aromatic: it is stable and the electrons are delocalized Compounds with 4n electrons are NOT aromatic (May be Antiaromatic) • Planar, cyclic conjugated molecules with 4n electrons are antiaromatic – They are much less stable than expected – They will distort out of plane and behave like ordinary alkenes cyclobutadiene cyclooctatetraene Which of the above is antiaromatic? • Cyclobutadiene – It has 4 electrons: 4n + 2 = 4, thus n = ½ (not an integer) – It is antiaromatic: The electrons are localized into two double bonds localized electrons • Cyclobutadiene – It has 4 electrons: 4n + 2 = 4, thus n = ½ (not an integer) – It is antiaromatic: The electrons are localized into two double bonds • It is so unstable that it dimerizes by a self-Diels-Alder reaction at low temperature dienophile diene • Cyclooctatetraene – It has 8 electrons: 4n + 2 = 8, thus n = 3/2 (not an integer) – It is nonaromatic: • the electrons are localized into four double bonds • it is tub-shaped not planar • it has four double bonds, reacting with Br2, KMnO4, and HCl as if it were four alkenes p orbitals not parallel for overlap Practice Problem: To be aromatic, a molecule must have 4n + 2 electrons and must have cyclic conjugation. Is cyclodecapentaene aromatic? • It has 10 electrons: 4n + 2 = 10, thus n = 2 (an integer) • It is not planar due to steric strain, thus the neighboring p orbitals are not properly aligned for overlap. It is not conjugated. Thus it is not aromatic. 6. Aromatic Ions • The Hückel 4n + 2 rule applies to ions as well as to neutral species: • To be aromatic, a molecule must be planar, cyclic conjugated system with 4n + 2 electrons • Example: Both the cyclopentadienyl anion and the cycloheptatrienyl cation are aromatic. • Example: Both the cyclopentadienyl anion and the cycloheptatrienyl cation are aromatic. The key feature of both is that they contain 6 electrons in a ring of continuous p orbitals Aromaticity of the Cyclopentadienyl Anion Not fully conjugated and not aromatic Unstable and nonaromatic Stable and aromatic • Cyclopentadiene is relatively acidic (pKa = 16) because its conjugate base, the aromatic cyclopentadienyl anion, is so stable. – Other hydrocarbons have pKa > 45 Aromaticity of the Cyclopentadienyl Anion • 1,3-Cyclopentadiene contains conjugated double bonds joined by a CH2 that blocks delocalization • Removal of H+ at the CH2 produces a cyclic 6-electron system, which is stable • Removal of H- or H• generate nonaromatic 4 and 5 electron systems • Relatively acidic (pKa = 16) because the anion is stable Aromaticity of the Cycloheptatrienyl Cation Not fully conjugated and not aromatic Stable and aromatic Unstable and nonaromatic • The cycloheptatrienyl cation (six electrons) is aromatic and very stable – Reaction of cycloheptatriene with Br2 yields cycloheptatrienylium bromide, an ionic substance containing the cycloheptatrienyl cation Aromaticity of the Cycloheptatrienyl Cation • Cycloheptatriene has 3 conjugated double bonds joined by a CH2 • Removal of H- at the CH2 produces the cycloheptatrienyl cation • The cation is a cyclic 6-electron system, which is stable and is aromatic • Removal of H+ or H• generate nonaromatic 7 and 8 electron systems Practice Problem: Draw the five resonance structures of the cyclopentadienyl anion. Are all carbon-carbon bonds equivalent? How many absorption lines would be in the 1H and 13C NMR spectra of the anion? Practice Problem: Cyclooctatetraene readily reacts with potassium metal to form the stable the cyclooctatetraene dianion, C2H82-. Why does the reaction occur so easily? What is the geometry of the dianion? 7. Aromatic Heterocycles: Pyridine and Pyrrole • A heterocycle is a cyclic compound that contains an atom or atoms other than carbon in its ring, such as N, O, S, P • There are many heterocyclic aromatic compounds and many are very common • Cyclic compounds that contain only carbon are called carbocycles (not homocycles) • Nomenclature is specialized • Example: Pyridine and Pyrrole Pyridine • Pyridine is a six-membered heterocycle with a nitrogen atom in its ring • electron structure resembles benzene (6 electrons) • The nitrogen lone pair electrons are in sp2 orbital, not part of the aromatic system (perpendicular orbital) • Pyridine is a relatively weak base compared to normal amines but protonation does not affect aromaticity Pyrrole • Pyrrole is a five-membered heterocycle with a nitrogen atom in its ring • electron system is similar to that of cyclopentadienyl anion • Four sp2-hybridized carbons with 4 p orbitals perpendicular to the ring and 4 p electrons Pyrrole • Nitrogen atom is sp2-hybridized, and lone pair of electrons occupies a p orbital (6 electrons) • Since lone pair electrons are in the aromatic ring, protonation destroys aromaticity, making pyrrole a very weak base Practice Problem: Thiophene, a sulfur-containing heterocycle, undergoes typical aromatic substitution reactions rather than addition reactions. Explain why thiophene is aromatic. It has 6 electrons: 4n + 2 = 6, thus n = 1 (an integer) It has a lone pair of electrons in a p orbital perpendicular to the plane Practice Problem: Draw an orbital picture of furan to show how the molecule is aromatic It has 6 electrons: 4n + 2 = 6, thus n = 1 (an integer) It has a lone pair of electrons in a p orbital perpendicular to the plane Practice Problem: Draw an orbital picture of imidazole, and account for its aromaticity. Which nitrogen atom is pyridine-like, and which is pyrrole-like? Which nitrogen atom is more electron-rich, and why? 8. Why 4n + 2? • According to the molecular orbital (MO) theory: – Cyclic conjugated molecules always have a single lowest-lying MO, above which the MOs come in degenerate pairs – When electrons fill the various molecular orbitals, it takes two electrons (one pair) to fill the lowestlying orbital and four electrons (two pairs) to fill each of n succeeding energy level • Only a total of 4n + 2 electrons fill bonding MOs Benzene • Benzene has its bonding orbitals filled (6 electrons) y1, the lowest-energy MO, is single and has two electrons y2 and y3, the next two lowest-energy MOs, are degenerate and have four electrons Cyclopentadienyl anion • Cyclopentadienyl anion has its bonding orbitals filled (6 electrons) y1 is single and has two electrons; y2 and y3 are degenerate and have four electrons - Cyclopentadienyl cation and radical do not have their bonding orbitals filled Practice Problem: Show the relative energy levels of the seven MOs of the cycloheptatrienyl system. Tell which of the seven orbitals are filled in the cation, radical, and anion, and account for the aromaticity of the cycloheptatrienyl cation 9. Polycyclic Aromatic Compounds: Naphthalene • Polycyclic aromatic compounds are – aromatic compounds with rings that share a set of carbon atoms (fused rings) – compounds from fused benzene or aromatic heterocycle rings carcinogenic (tobacco) Characteristics of Polycyclic Aromatic Compounds • They are cyclic, planar and conjugated molecules • They are unusually stable • They react with electrophiles to give substitution products, in which cyclic conjugation is retained, rather than electrophilic addition products • They can be represented by different resonance forms • They have 4n + 2 electrons, delocalized over the ring Naphthalene • Naphthalene has three resonance forms • Naphthalene reacts slowly with electrophiles to give substitution products Naphthalene • Naphthalene is a cyclic, conjugated electron system, with p orbital overlap both around the ten-carbon periphery of the molecule and across the central bond – It has ten delocalized electrons (Hückel number) Practice Problem: Azulene is an isomer of naphthalene. Is azulene aromatic? Draw a second resonance form of azulene. Practice Problem: Naphthalene is sometimes represented with circles in each ring to represent aromaticity. How many electrons are in each circle? 10. Spectroscopy of Aromatic Compounds Aromatic compounds can be identified by: – Infrared (IR) Spectroscopy – Ultraviolet (UV) Spectroscopy – Nuclear Magnetic Resonance (NMR) Spectroscopy Infrared Spectroscopy • Aromatic rings have C–H stretching at 3030 cm1 and peaks in the range of 1450 to 1600 cm1 • Substitution pattern of the aromatic ring: Monosubstituted: 690-710 cm-1 730-770 cm-1 o-Disubstituted: 735-770 cm-1 m-Disubstituted: 690-710 cm-1 810-850 cm-1 p-Disubstituted: 810-840 cm-1 • Example: Toluene (IR) 3030 cm1 Monosubstituted: 690-710 cm-1 730-770 cm-1 Ultraviolet Spectroscopy • Aromatic rings have peaks near 205 nm and a less intense peak in 255-275 nm range – Aromatic compounds are detectable by UV spectroscopy since they have a conjugated electron system Nuclear Magnetic Resonance Spectroscopy 1H NMR: – Aromatic H’s are strongly deshielded by ring and absorb between 6.5 and 8.0 – Peak pattern is characteristic positions of substituents • Ring Current is a property unique to aromatic rings – When aromatic ring is oriented perpendicular to a strong magnetic field, delocalized electrons circulate producing a small local magnetic field – This opposes applied field in middle of ring but reinforces applied field outside of ring • Ring Current accounts for the downfield shift of aromatic ring protons in the 1H NMR spectrum – Aromatic 1H’s experience an effective magnetic field greater than applied – It results in outside H’s resonance at lower field • Ring Current produces different effects inside and outside the ring – Outside 1H are deshielded and absorb at a lower field – Inside 1H are shielded and absorb at a higher field • Ring Current is characteristic of all Hückel aromatic compounds – Aryl 1H absorb between 6.5-8.0 – Benzylic 1H absorb between 2.3-3 downfield from other alkane 1H • Example: p-bromotoluene (1H NMR) The 4 aryl protons: Two doublets at 7.02 and 7.45 The benzylic CH3 protons: a singlet at 2.29 Integration 2:2:3 13C NMR – Carbons in aromatic ring absorb between 110 to 140 – Shift is distinct from alkane carbons but is in same range as alkene carbons Chapter 15