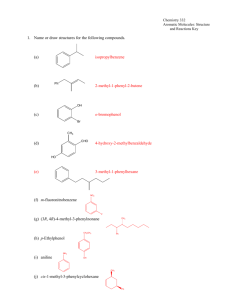
Aromatic Compounds
advertisement

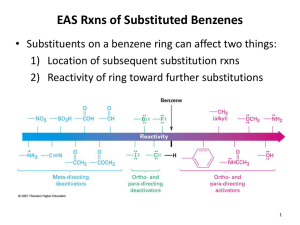
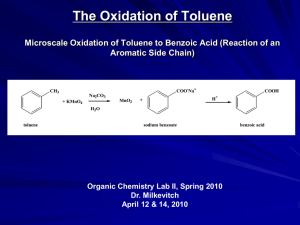
Aromatic Compounds Nature presents us with a wide array of naturally occurring substances. Some structural subtypes occur with high frequency among the millions of know naturally occurring substances. One frequently occurring structural subtype is a six-membered ring with three double bonds. This subtype has been extensively explored over the past 150 years, and found to possess unusual stability. It is believed that this stability is due to a particular property of possessing a closed circle of pi orbitals possessing six pi electrons. As we shall see, these cyclic, unsaturated systems seem to possess some unusual chemical stability. More examples of such stabilized cyclic systems are shown below. Pyrrole Furan Aromatic Systems are Characterized by Their Chemical Stability • Note the chemical stability of the aromatic systems to the reaction conditions in the following slides Note aromatic system’s stability toward hydrogenation Note aromatic systems’ stability toward strong reducing agent LiAlH4 Note the (two) aromatic systems’ stability toward Br2 Note the aryl iodide’s stability toward SN2 substitution (SN2 substitution occurs at the sp3 hybridized carbon) Note the aryl iodide’s stability toward the strong base (potassium tert-butoxide) used to effect elimination It is important to understand that, in heterocyclic ring systems, the lone pair of electrons on the heteroatom may be required as part of the aromatic sextet, in which case, the heteroatom is not basic nor nucleophilic. In the case of pyrrole, the nitrogen is not basic nor nucleophilic, since the nitrogen lone pair is part of the aromatic sextet. Or, it may be that the lone pair of the heteroatom is not required for the aromatic sextet, in which case the heteroatom may be basic and nucleophilic. In the case of pyridine, above, the lone pair is not a part of the aromatic sextet, and is basic and nucleophilic. Another important system is imidazole, shown below, the heterocyclic system of the amino acid histidine. One of the nitrogen atoms is basic, while the other is not. Reactions of Aromatic Systems: Electrophilic Aromatic Substitution Notice that cyclohexene (right, green box) is quite reactive toward strong acids, bromine, and strong oxidizing agents. Under these same conditions, benzene, blue box to right, does not react. However, benzene can be made to react under forcing conditions shown at left. But the products (from benzene reaction) are different from what one might expect, using the reactivity of cyclohexene as a predictive model. Mechanism of Electrophilic Aromatic Substitution by attack of electrophile (E+) on the benzene ring The Friedel-Crafts Reaction Sometimes, substituents on the aromatic ring may ‘direct’ the incoming electrophile to attack specific carbon atoms of the aromatic ring, as shown in the following examples. Notice that substituents that stabilize an adjacent carbocation (either by resonance or by electronegativity) activate the aromatic ring toward electrophilic substitution. Notice that electron-withdrawing groups deactivate the ring toward electrophilic substitution (reduce its reactivity toward electrophiles). Nucleophilic Substitution at the Benzene Ring Recall that nucleophilic substitution at sp3 hybridized carbon usually occurs much more rapidly (loss of the benzylic chloride) than substitution an (sp2-hybridized) aryl halide itself, as shown in the example below. But, in certain very specific conditions, substitution of an aryl halide can occur. The two most common mechanisms for substitution of an aryl halide are: •The Benzyne Mechanism (under strongly basic conditions) and • The Addition-Elimination Mechanism (when the aryl-halide has electron-withdrawing groups oriented ortho- and para- to the halide. Treatment of Aryl Halides with Extremely strong bases (amide anions, NaNH2, pKa of ammonia = 38) can cause substitution reactions… (note that the above table shows the conjugate acids only) But the mechanism involves a two-step process of elimination-addition. Treatment of aryl halides having strongly electronwithdrawing substituents (at the 2- and the 4-position) can also cause substitution reactions But… The mechanism involves addition-elimination. Reactions of Side Chains and Attached Functionality on Aromatic Compounds Recall that aryl nitro compounds are readily available by electrophilic substitution, using nitric acid. These product aryl-nitro compounds are synthetically useful, since the nitro group can be easily reduced to an amino group. Likewise aryl amino compounds are synthetically valuable, since the NH2 group can be transformed into an aryl diazonium salt, which is a useful intermediate for substitution at an aromatic carbon. Treatment of amines with nitrous acid (HONO) generates diazonium salts Aryl Diazonium salts are useful in substitution reactions The Sandmeyer Reaction Swiss chemist (1854-1922) after whom reaction is named Benzylic Positions Can be Selectively Oxidized (all the way to the carboxylic acid) by Potassium Permanganate The benzylic position can be readily halogenated via a free-radical process, as shown below. The Clemmensen Reduction Hydrogenolysis of Benzyl Esters and Benzyl Ethers It is possible to reduce a benzene ring to a 1,4-cyclohexadiene, using a reduction protocol known as the Birch Reduction The mechanism of the Birch Reduction involves successive one electron transfers as shown below. The alkali metal serves as a source of electrons. The solvent is usually liquid ammonia.