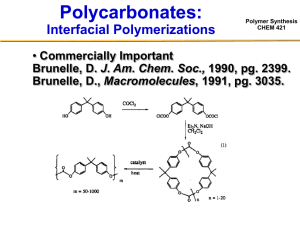
Polymer Synthesis CHEM 421
advertisement

Polymer Synthesis CHEM 421 • Chapter 3.9 (Odian) Free Radically Polymerized Monomers Polymer Synthesis CHEM 421 Thermodynamics of Chain Polymerizations • Relief of “strain” is the driving force –Exothermic process Polymer Synthesis CHEM 421 Thermodynamics • ΔG, ΔH, and ΔS ΔG = ΔH - T ΔS • Chain polymerizations: » Enthalpy »Exothermic (- ΔH) » Entropy »Negative ΔS • Polymer favored from enthalpic considerations but un-favored from entropic considerations Polymer Synthesis CHEM 421 Thermodynamics Polymer Synthesis CHEM 421 • ΔHpolymerization depends upon differences in: –Resonance stabilization of polymer versus the monomer –Steric strain in monomer versus the polymer –Hydrogen bonding or dipolar interaction in monomer versus the polymer Thermodynamic Trends (kJ/mole) (J/°K-mole) Polymer Synthesis CHEM 421 • Ethylene Styrene α-Methyl styrene or • Ethylene methyl acrylate methyl methacrylate • PTFE (!) – Most exothermic polymerization known Equilibrium Considerations Polymer Synthesis CHEM 421 • For most polymerizations, there is a temperature where the reaction becomes reversible • The position for the monomer / polymer equilibrium kp kdp will be dependent on the temperature • ΔG = ΔH - T ΔS – Polymerization: – De-polymerization: ΔS = ΔS = • With increasing temperature the equilibrium will shift? Equilibrium Considerations • When Rp = Rdp – Ceiling temperature Polymer Synthesis CHEM 421 Thermodynamics Polymer Synthesis CHEM 421 • The reaction isotherm: ΔG = ΔG° + RTlnK is applicable. Where ΔG° is the ΔG of polymerization for the monomer and the polymer in the appropriate standard states –Monomer Std. State: pure liquid –Polymer Std. State: crystalline state if possible, otherwise amorphous state Thermodynamics Polymer Synthesis CHEM 421 • At equilibrium, ΔG = 0 by definition: ΔG° = - RTlnK Equilibrium constant is defined by Keq = kp / kdp [Mn+1•] 1 Keq = ————— = ——— [Mn•] [M] [M] Thermodynamics Polymer Synthesis CHEM 421 • Combine: ΔH° Tc = ————— ΔS° + Rln[M]c [M]c is the equilibrium monomer concentration as a function of reaction temperature –The monomer concentration in equilibrium with the polymer increases as the temperature increases Ceiling Temperature • Poly(α-methyl styrene) –Tg = 170 °C –Tceil = 61 °C • Processing temperature? Polymer Synthesis CHEM 421 Ceiling Temperature Polymer Synthesis CHEM 421 • Poly(methyl methacrylate) –Tg = 125 °C –Tceil = 164 °C • Recycle… PMMA 300 °C 99% MMA monomer Heats of Polymerization and Ceiling Temperatures Monomer Polymer Synthesis CHEM 421 Heat of Polymerization (kcal/mol) -16 -7 Ceiling Temperature (°C) 235 61 Methyl acrylate Methyl methacrylate -20 -13 ---164 Ethylene Propylene Isobutene -26 -21 -17 407 300 50 Styrene α-Methyl styrene Polymerization Processes Polymer Synthesis CHEM 421 • Solution Polymerization • Bulk (Mass) Polymerization • Heterogeneous Polymerizations Solution Polymerizations Polymer Synthesis CHEM 421 • Ingredients –Monomer –Solvent –Initiator Rp = kp [M] (kd f [I] / kt)1/2 Rp Rp kp [M] [M•] kp [M] ٧ = — = — = ———2 = ————— Ri Rt 2 (kt kd f [I])1/2 2 kt [M•] Solution Polymerization M M I M M I M M I M I M M I Solvent M • • • • hν or Δ Polymer Synthesis CHEM 421 P M P M I I P P M M P P I P Solvent P Solvent, monomer & initiator Polymer remains soluble in the solvent Easy temperature, viscosity, MW control Free radical kinetics apply Solution Polymerization Polymer Synthesis CHEM 421 • Considerations: –Chain transfer to solvent –Purity of polymer (difficulty in removing solvent) • Used for: vinyl acetate, acrylonitrile, and esters of acrylic acid Bulk (Mass) Polymerizations Polymer Synthesis CHEM 421 • Ingredients: Monomer and Initiator only • Kinetics follows solution polymerization kinetics…Rp and ٧ Bulk (Mass) Polymerization Polymer Synthesis CHEM 421 • Considerations: –Hard to control: high activation energies, gel effect –Equipment: elaborate, strong stirring due to viscosity increase –Temperature Control: local hot spots » Can lead to degradation, discoloration, and broad MW distribution » “Runaway” reactions –Used for styrene and methyl methacrylate (Chain Growth) » Low conversion and separation/recycling of un-reacted monomer