Transition Metal Chemistry - WordPress.com

Transition Metal
Chemistry
The Chemistry of the d-block elements
s
The Periodic Table
p d
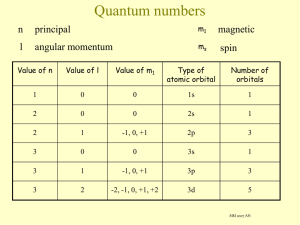
Electronic configurations
Using the aufbau principle :
1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 2 , 3d 1
1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 2 , 3d 10
1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 2 , 3d 6
The two exceptions
You would expect Chromium to have the electronic configuration:
1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 2 , 3d 4
But in fact it has the configuration: 1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 1 , 3d 5
There is a special stability associated with half-filled and full subshells.
Copper: 1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 1 , 3d 10
Ions
• Transition metals are defined as metallic elements with an incomplete d sub-shell in at least one of their ions.
• Form positive (+) ions by losing electrons.
• These electrons come from the 4s sub-shell first, then from the 3d sub-shell:
Fe atom: 1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 4s 2 , 3d 6
Fe 2+ ion: 1s 2 , 2s 2 , 2p 6 , 3s 2 , 3p 6 , 3d 6
Complex Ions and Complexes
Understanding Transition metal compounds
In aqueous solution
• Transition metal ions exist as complex ions in aqueous solution, e.g.
Co(H
2
O)
6
2+
What shape is this?
Ligands
Formed through donation of electron pairs
Coordinate covalent bond
The water molecules are an example of ligands
LIGAND : molecules or anions which attach to the metal atom in a complex via coordinate (dative) covalent bonds
Number of bonds formed with ligands = COORDINATION NUMBER
Assemble is known as a COMPLEX ION
So instead of existing in solution as free ions, e.g. Cu 2+ , exist as complex ions, Cu(H
2
O)
5
2+
Shapes of complex ions
Naming ligands
Ligand
Bromide
Carbonate
Cyanide
Hydroxide
Ammonia
Carbon monoxide
Water
Name
Bromo
Carbonato
Cyano
Hydroxo
Ammine
Carbonyl
Aqua
Categories of ligand
• MONODENTATE LIGAND: the ligand bonds to the metal using one atom
• POLYDENTATE LIGANDS: the ligand bonds to the metal using more than one atom
Common Examples
Chlorophyll Haemoglobin
Determining coordination number
Number of bonds formed with ligands = COORDINATION NUMBER
Complex ion
Ag(NH
3
)
2
+
HgI
3
-
W(CO)
6
4+
Coordination number
6 is the most common coordination number
2
3
6
Forming a complex
• The cation or anion cannot exist on their own and must have their charges balanced
• The complex ion will bond with oppositely charged ions to form a complex
Complex
[Pt(NH
3
)
6
]Cl
4
K
4
[Fe(CN)
6
]
Cation
Pt(NH
3
)
6
4+
[Pt(NH
3
)
4
Cl
2
]Cl
2
Pt(NH
3
)
4
Cl
2
2+
K +
Anion
Cl
Cl
-
-
Fe(CN)
6
4-
Naming Coordination
Compounds
1. Cation precedes anion
2. Complex ion names are one word: ligands first, then metal
3. Ligands will have a Greek prefix in front
4. If the complex ion is an anion, it ends in -ate
5. The metal name is followed by the oxidation state in Roman Numerals
Old vs New
• CuSO
4
·5H
2
O vs [Cu(H
2
O)
5
SO
4
]
• Copper(II) sulphate pentahydrate
• Pentaaquacuprum(II) sulphate
Colour in
Transition Metal
Compounds
Why coloured?
Transition metal ions are often coloured
They absorb EM radiation because of loss of degeneracy of d-orbitals
Those which absorb in the visible region will appear the complementary colour
• The 5 d-orbitals in an isolated atom are degenerate
• Ligands cause the d-orbitals to become nondegenerate
• Different ligands cause different splitting effects
Crystal field splitting
(energy), Δ
In an octahedral complex , the ligands lie on the x, y and z axis
Their electrons have a greater repulsive effect on the d-orbitals which lie on the same axis
Spectrochemical Series
• An arrangement of ligands according to the relative magnitudes of the crystal field splittings they induce in the d-orbitals of a metal ion
Weak-bonding ligands
• I < Br < Cl < F < OH < H
2
Strong-bonding ligands
O < NH
3
<
NO
2
< CN < CO
Increasing Δ
Therefore, different ligands will result in different colours
Gemstones
Cr 3+ in Al
2
O
3
Cr 3+ in Be
3
Al
2
(SiO
3
)
6
Catalysis
Transition metals as catalysts
• Catalysts provide an alternative pathway with a lower activation energy
• Transition metals can use half-filled or empty orbitals to form intermediate complexes (e.g.
4p)
• They can change oxidation state during a reaction, then revert back to their original state