EDTA Titration of Water
advertisement

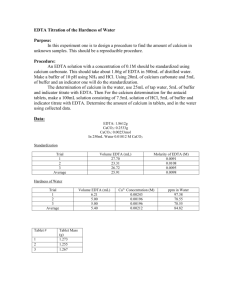
Presented By: Jared Poist and Monique Gray Hard Water Hard water is a detectable amount of dissolved cations in water. Drinking hard water poses no known health risk. It causes water to taste bad. One of the main problems with hard water is it leaves behind residues and precipitants on a majority of surfaces. EDTA Ethylenediaminetetracetic Acid This chemical compound forms 1:1 metal complexes with the majority of the metal cations. EDTA plays a strong role as a metal-binding agent in industrial processes, products/supplies such as cleaning agents and food additives that prevent metalcatalyzed oxidation of food. Biochemistry Application EDTA is used in biochemistry to stop enzymatic activity by binding with the divalent cations from the active site of enzymes, thus, preventing it from functioning. Common divalent cations are Ca2+ and Mg2+. EDTA Continued EDTA is a mutlidentate ligand or a chelating ligand The chelate effect is the ability of multidentate ligands to form more stable metal complexes than those formed by similar monodentate ligands. The more electrons pairs an atom has available for binding the more stable the complex will be. Compleximetric Titrations Basically, titrations that are based upon forming metalcation complexes with different ligands. EDTA Acid-Base Titration Water sample Information Deer Park Spring Water Filtered Water (from Jared’s home refrigerator ) Tap Water Data (came from previous lab experiment) Introduction to Our Experiment Plan: use the EDTA titration technique to find the hardness of water for filtered water and spring water. Hypothesis: (1) The hardness in tap water should be higher than both filtered water and spring water. (2) The hardness in filtered water should be higher than spring water Procedure: Standardizing EDTA Prepared Solutions: 0.1 CaCl2 solution and 0.01 EDTA Solution Added 3mL of CaCl2 into a 250mL flask, 5mL ammonia buffer (pH 10), and 5 drops of calmagite indicator Standardized EDTA and recorded 3 good trials Standardizing EDTA Results Trial # EDTA Added (mL) Molarity of EDTA 1 15.20 mL 0.01973 M 2 14.0 mL 0.02140 M 3 15.0 mL 0.02000 M Average Molarity: 0.0204 M Procedure Continued (1) Calibrated a pH meter with standard solutions, and used the pH meter to make a buffer with pH 10. (2) Added 3mL ammonia buffer, 25mL water (sample), and 5 drops indicator. (3) Three good trials for each water sample. Calculations mL CaCl2 added x 0.100 Ca2+ M / 1000 mL x 1mol EDTA/ 1 mol Ca2+ x 1 / L EDTA added = Molarity of EDTA L of EDTA x Average Molarity x 1 mol Ca2+/1 mol EDTA x 1/ 0.025L = concentration of hardness Ca2+ Molarity x grams Ca2+/ 1 mol x 1000mg/1g = ppm EDTA Titration Results Trial # Spring Water Amount EDTA Added mL 1 25mL 1.20 mL 2 25mL 1.20 mL 3 25mL 1.10 mL Trial # Filtered water Amount EDTA Added mL 1 25 mL 1.9 mL 2 25 mL 1.7 mL 3 25mL 2.01 mL EDTA Titration Results Trial # 1 Amount Tap Water mL Amount EDTA Added mL 1 25 mL 2.68 mL 2 25 mL 2.31 mL 3 25 mL 2.25 mL Hardness of Water Samples Results Spring Water Trial # Ca2+ Molarity ppm 1 0.0009792 39.244 2 0.0009792 39.244 3 0.0008976 35.974 Filtered Water Trial # Ca2+ Molarity ppm 1 0.0015504 62.1370 2 0.00138720 55.5970 3 0.00164016 65.7343 Hardness of Water Sample Results Tap Water Trial Ca2+ Molarity ppm 1 0.00236 94.584 2 0.00203 81.358 3 0.00198 79.354 Conclusion We found that our first hypothesis was correct, which stated that tap water had a greater hardness than spring and filtered water. We found that our second hypothesis was true, which stated that filtered water would have a higher hardness than spring water.