Real World Presentation
advertisement

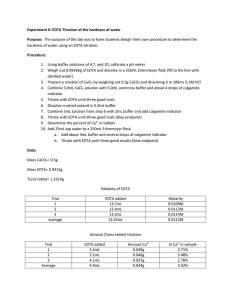
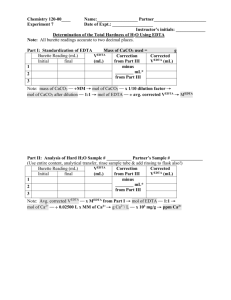
Kirah Howell, Margaret Walker & Melanie Fannon To determine the pH of five water samples To determine total hardness of these samples To test for how much sulfate is in the water samples Water should have a pH between 6.5 and 8.5 Hard water causes problems to water Too much sulfate can affect water Calibrate pH meter with 4,7 & 10 buffers Pour 25 mL of water sample into beaker Test each sample Make 2L of 0.005M EDTA solution Make 200 mL of CaCO3 solution Standardize EDTA Combine 3 mL of CaCO3, 5 mL of ammonia buffer, few drops of calmagite indicator & 25 mL of water sample into 250 mL Erlenmeyer flask Titrate with EDTA Make BaCl2 solution Combine 150 mL of water sample , excess of BaCl2 solution and a few drops of concentrated HCl in a 300 mL beaker and boil a hour then cool for 2 days Filter samples Titrate with EDTA solution Low Molarity of the EDTA Indicator Multiple times to find the end point of each sample. pH Creek 7.72 Little Run 7.82 Business Building 7.70 Campbell Hall 8.12 Country Club 7.78 [Ca2+] and hardness Location Vol (mL) EDTA Business Building 38.30 Country Club 34.80 Campbell Hall 34.90 Little Run 35.20 Creek 35.03 [Ca2+] and hardness Location [Ca2+] Hardness (ppm) Business Building .00766 306.99748 Country Club .00696 252.2762133 Campbell Hall .00698 279.74444 Little Run .00704 282.14912 Creek .00674 270.12572 SO42Location Vol (mL) EDTA Business Buidling 86.67* Country Club 41.10 Campbell Hall 39.53 Little Run 40.73 Creek 41.10 SO42Location [SO42-] Ppm Business Building* 7.153571016x10-5 6.871791854 Country Club 1.265206813x10-4 12.15370316 Campbell Hall 1.16367316x10-4 11.17836074 Little Run 1.203044439x10-4 11.55656519 Creek 1.16367316x10-4 11.17836074 The Experiment was successful The differences of each site Challenges that were faced Errors that we encountered Belle-Oudry, D. Quantitative Analysis of Sulfate in Water by Indirect EDTA Titration. Journal of Chemical Education. [Online] September 1, 2008. DOI: 10.1021/ed085p1269. http://pubs.acs.org/doi/abs/10.1021/ed085p1269 (accessed December 6, 2013) EPA (Environmental Protection Agency)