Empirical Formulas
advertisement

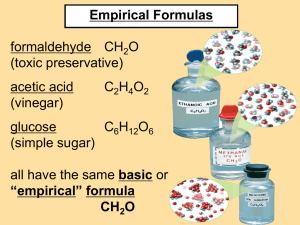
What Could It Be? Empirical Formulas The empirical formula is the simplest whole number ratio of the atoms of each element in a compound. Note: it is not necessarily the true formula of the compound. Example: The molecular formula for glucose is C6H12O6, but its empirical formula is CH2O It’s kind of like a fraction reduced to its lowest terms. Molecular Formulas The molecular formula gives the actual numbers of each element, and thereby represents the true formula of the compound. Calculating the Empirical Formula Example 1: A compound is found to contain the following… 2.199 g Copper 0.277 g Oxygen Calculate it’s empirical formula. Calculating the Empirical Formula Step 1: Convert the masses to moles. Copper: 1 m oleCu 2.199g Cu 0.03460 m olesCu 63.5 g Cu Oxygen: 1 m ole O 0.277g O 0.01731m oles O 16.0 g O Calculating the Empirical Formula Step 2: Divide all the moles by the smallest value. This gives the “mole ratio” 0.03460 mol Cu 1.999 Cu 0.0173mol 0.0173 molO 1.000O 0.0173 mol Calculating the Empirical Formula Step 3: Round off these numbers, they become the subscripts for the elements. Cu2O Example 2: A material is found to be composed of 38.7% Carbon, 51.6% Oxygen, and 9.7% Hydrogen. By other means, it is known that the molecular weight is 62.0 g/mol Calculate the empirical and molecular formula for the compound. If you assume a sample weight of 100grams, then the percents are really grams. Example 2: A material is found to be composed of 38.7% Carbon, 51.6% Oxygen, and 9.7% Hydrogen. By other means, it is known that the molecular weight is 62.0 Calculate the empirical and molecular formula for the compound. 1m ole 3.23m ol Carbon: 38.7 gram s 12.0 gram s Oxygen: Hydrogen: 1 m ole 51.6 gram s 3.23m ol 16.0 gram s 1 m ole 9.7 gram s 9.7m ol 1.0 gram s Example 2: A material is found to be composed of 38.7% Carbon, 51.6% Oxygen, and 9.7% Hydrogen. By other means, it is known that the molecular weight is 62.0 Calculate the empirical and molecular formula for the compound. 1m ole 3.23m ol Carbon: 38.7 gram s 1.00 12.0 gram s 3.23 Now, divide all the moles by the smallest one, 3.23 Oxygen: 1 m ole 51.6 gram s 3.23m ol 1.00 16.0 gram s 3.23 Hydrogen: 1 m ole 9.7 gram s 9.7m ol 3.00 1.0 gram s 3.23 Example 2: A material is found to be composed of 38.7% Carbon, 51.6% Oxygen, and 9.7% Hydrogen. By other means, it is known that the molecular weight is 62.0 Calculate the empirical and molecular formula for the compound. So, the empirical formula must be CH3O The molecular weight of the empirical formula is…. C 12 x 1 = 12 g/mol H 1x3 = 3 g/mol O 16 x 1 = 16 g/mol 31 g/mol Remember, the empirical formula is not necessarily the molecular formula! MW of the empirical formula = 31 MW of the molecular formula = 62 Molecular Multiplying Factor Em pirical 62 2 31 2x Empirical Formula ( CH3O ) = C2H6O2 Molecular Formula Remember, the molecular formula represents the actual formula. What if the mole ratios don’t come out even? Example 3: A compound is analyzed and found to contain 2.42g aluminum and 2.15g oxygen. Calculate its empirical formula. 1m ol Aluminum: 2.42g .0896m ol 1.0 X 2 = 2 27g .0896 Oxygen: 1m ol 2.15g .134m ol 1.495 X 2 = 3 16g .0896 Al2O3