Proteomic analysis of bile in pancreatic adenocarcinoma
advertisement

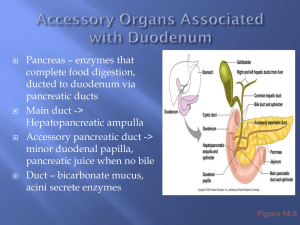
ACADEMIC TRAINEES ANNUAL EVENT 2011 NEUTROPHIL GELATINASE-ASSOCIATED LIPOCALIN IS ELEVATED IN BILE FROM PATIENTS WITH MALIGNANT PANCREATOBILIARY DISEASE A. Zabron1, V. Horneffer-van der Sluis 2, C. Wadsworth 1, M. Gierula 2, A. Thillainayagam 3, P. Vlavianos3, D. Westaby 3, S. Taylor-Robinson 1, R. Edwards 2, S. Khan 1 1Hepatology and Gastroenterology Section, Division of Diabetes Endocrinology and Metabolism, Department Medicine, 2Centre of Pharmacology and Therapeutics , Imperial College London, 3Gastroenterology Unit , Hammersmith Hospital, Imperial College Healthcare NHS Trust, London, United Kingdom Previously presented at UEGW and BSG. Accepted for publication in the American Journal of Gastroenterology sdfgafgafga of Content • Focus on pancreatic adenocarcinoma • Significance and current diagnostic difficulties • Proteomic technique • Results • Possible clinical use and further work • Summary Pancreatic adenocarcinoma • Imperial AHSC HPB tertiary referral centre • 10th commonest cancer in the UK • Mortality similar to incidence • Diagnosis by cross-sectional imaging, histology / cytology and serum biomarkers imperfect • Frequently present with biliary obstruction Anatomy of biliary system Endoscopic retrograde cholangiopancreatography : ERCP Stenting across obstruction via ERCP Bile as a diagnostic sample in malignant pancreatobiliary disease • Current cytological analysis of brushings – low sensitivity • Spy-glass • Metabonomic interest • Recent proteomic interest IGF1 and pancreatic elastase-3B higher in CC bile (Alvaro D. et al., 2009) CEACAM-6 and MUC1(CA 19-9) higher in PA bile (Farina et al., 2009) Current Study Aims: To investigate the potential of bile as a source of novel biomarkers in pancreatic adenocarcinoma Design: Cohort study Discovery phase - label-free proteomics to unfractionated sample, antibody-based techniques Validation cohort Sample collection and preparation • Bile aspirated at ERCP in patients with benign or malignant disease prior to introduction of contrast • Routine blood results/ serum markers, demographics etc recorded • Blood and urine collected at same event Label-free proteomics: tryptic peptide production Crude centrifuged sample: water/ lipids/ bile acids and pigments/ proteins No cancer Pancreatic cancer Slice gel into equal fragments containing denatured proteins in size ranges SDS-PAGE Tryptic peptides In-gel protease digestion Extract from gel slice Label-free proteomics: LC-MS/MS Intensity of detection Mass/carge ration (m/z) Peptide fragments Retention time (min) Retention time (min) Retention time (min) Benign Retention time (min) 11 CC Retention time (min) Mass/charge ration (m/z) 1 Mass/charge ration (m/z) Mass/charge ration (m/z) Label-free proteomics: Normalisation and Quantification 1 8 PaCa VPLQQNFQDNQFQGK Retention time (min) “196,454.71” Progenesis, nonlinear dynamics Normalised abundance profile of single peptide spot Normalised abundance of a specific protein in gel piece Normalised abundance volume of all peptide spots of an identified protein Data analysis • SEQUEST » human RefSeq protein sequence database (NCBI) • Progenesis » Quantification by non-linear dynamics • Statistical analysis Results of label-free proteomics •Bile analysed from patients with pancreatic adenocarcinoma (n=4) and benign biliary disease (n=4) •Over 200 different proteins identified including S100A6, LCN2, CEACAM6, REG1α, PRDX6 •10 proteins varied significantly in abundance between benign and malignant groups Volcano plot of identified proteins Volcano plot of identified proteins NGAL_HUMAN: Lipocalin 2 or Neutrophil gelatinaseassociated lipocalin Normalised abundance volume of all peptide spots of lipocalin 2 •P = 0.029, protein fold change 13.9 •10 unique tryptic peptides •52% protein sequence coverage NGAL as a novel biomarker: Biological plausibility • “stress protein” • Novel urinary biomarker in for acute kidney injury • Overexpression in malignancy e.g. breast, ovarian, colon and pancreatic tissue Immunoblotting : Clinical characteristics Benign Malignant 22 16 Age [year] 59.83 ± 20.84 71.08 ± 11.62 0.043 Albumin [g/l] 33.84 ± 7.59 27.25 ± 7.7 0.013 Bilirubin [mmol/l] 35.33 ± 96.23 210.08 ± 155.3 <0.001 ALT [IU/l] 83.7 ± 101.8 298.17 ± 248.7 <0.001 ALP [IU/l] 346.8 ± 494.2 768.00 ± 602.9 0.006 CRP 15.14 ± 20.23 96.83 ± 100.0 0.008 urea 6.305 ± 4.116 5.59 ± 2.3 NS creatinine 97.40 ± 45.17 86.86 ± 33.4 NS Number p-value Western blot of NGAL in bile Gel 1 Gel 2 P<0.0001 ELISA of NGAL in different body fluids b) p=0.007 NS c) NS 180 1800 160 600 140 NGAL [ng/ml] 500 NGAL [ng/ml] a) 400 300 200 100 80 60 40 100 20 0 0 Benign Bile 120 M alignant Serum Benign M alignant Urine NGAL in bile as a diagnostic marker Receiver Operator Characteristics Area Under the Curve: 0.80 Cut-off level [ng/ml] Specificity% Sensitivity % < 569.5 54.29 100.0 < 661.0 54.29 92.31 < 803.8 54.29 84.62 < 952.5 57.14 84.62 < 1015 60.00 84.62 < 1080 62.86 84.62 < 1142 62.86 76.92 < 1230 65.71 76.92 < 1319 68.57 76.92 < 1419 68.57 69.23 < 1549 71.43 69.23 Combination of NGAL in bile and serum CA19-9 Cut-off NGAL in bile: 3000 ng/ml Cut-off serum CA19-9: 110 U/ml Below cut-off Above cut-off Benign 13 3 Malignant 2 11 Sensitivity: Specificity: PPV: NPV: 85% 82% 79% 87% Validation cohort Benign (n=14) Malignant (n=7) Diagnoses (n) Stone (6), Chronic Pancreatitis (3), PSC (3), Leak (2) Pancreatic Adenocarcinoma (5), Gallbladder Cancer (1), HCC (1) Female:male 5:9 4:3 Age [year] 61 (36-79) 75 (64-78) NS Albumin [g/l] 35.5 (27.5-42.0) 31 (26-36) NS Bilirubin [mmol/l] 15.0 (6.5-26.0) 121 (14-421) 0.004 ALT [IU/l] 60.0 (28.0-156.5) 168 (126-357) NS ALP [IU/l] 181 (122-1176) NS 620 (350-917) p-value Cut-off >570ng/ml gives sensitivity 100%, specificity 55% Biological functions of NGAL in malignancy Schematic model of NGAL-mediated iron traffic. [Kai M. Schmidt-Ott et al. J Am Soc Nephrol 18: 407–413, 2007] A) B) Principal cellular mechanisms attributed to NGAL in determining its pro- and anti-tumoral effects. General structure of A) the ferrichromes, prototypical hydroxamate type siderophores, and B) ferric enterobactin, a prototypical catechol-type siderophore. [Neilands J B, et al. J. Biol. Chem. 1995;270:26723-26726] [Bolignano D, et al. Cancer Lett. (2010) Vol. 288(1): pp 10-6.] Summary • Valuable HPB patient cohort and scientific facilities available at Imperial AHSC • This study identified over 200 proteins in bile • Confirmed that proteomic analysis of body fluids allows identification of potential biomarkers in pancreatobiliary malignancy • There are significant differences in the proteome of bile in malignant and benign pancreatic disease Summary 2 • This study highlights the increased abundance of bile LCN2/ NGAL in pancreatic adenocarcinoma. • Further work is required to elucidate the possible use of biliary LCN2/NGAL as a clinical marker of disease. Future work • Increasing collaborations with Imperial HPB teams to expand recruitment • Validation of biliary NGAL as a diagnostic and/ or prognostic marker in pancreatic cancer • Exploration of its biological mechanism e.g. role stabilising MMP-9 • Extension of this approach to other biliary tract disease Acknowledgements Patients! HPB and Endoscopy teams at Imperial AHSC Proteomics team at Imperial College British Liver Trust Alan Morement Memorial Fund Thank you Any questions?