B5_Pictures of the Mind - NU-TEACH

BELLRINGER
What is the chemical formula for water? Draw what one molecule of water looks like and draw what 5 molecules of water looks like.
1 Molecule H
2
0 5 Molecules H
2
0
Pictures of the Mind
Macroscopic vs. Microscopic
We live in a macroscopic world.
Atoms and elements exist in a microscopic world.
How do atoms in tin foil look or what do atoms in water look like?
Create models or representations to help us understand things.
Anatomy Models
Architecture Models
Biology Models
Model Models?
Chemistry Models
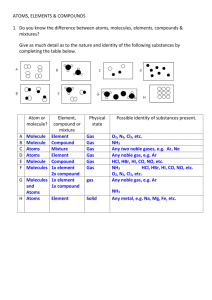
Sample Problem # 1
• Draw a model of a homogenous mixture composed of three different gaseous elements.
– Homogenous: Uniform throughout (compound should be intermingled and evenly distributed)
– Can the atoms of the elements exist as diatomic molecules?
Answer
Sample Problem # 2
• What kind of matter does the following model represent? Explain your answer!
Answer
• This model represents a heterogenous mixture of two molecular substances, each of which consists of two types of atoms.
Sample Problem # 3
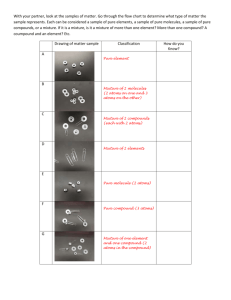
• Draw a model of a container of each of the following samples of matter.
– Table 1 and Table 7: Mixture of gaseous elements X and Z.
– Table 2 and Table 6: A two-atom compound of X and
Z.
– Table 3 and Table 5: A four atom compound of X and Z
– Table 4: A solution composed of a solvent that is a two-atom compound of L and R, and a solute that is a compound composed of two atoms of element D and one atom of element T.
Table 1 & Table 7
Table 2 & Table 6
Table 3 & Table 5
Table 4
Sample Problem # 4
• Answer Question #4 on your handout!
Sample Problem # 5
• The element Iodine (I) has a greater density in its solid state than its gaseous state. Draw models that depict and account for this difference at the particulate (really small!) scale. Iodine normally exists as a two-atom molecule.
Answer # 5
Sample Problem # 5
• A chemistry student at Riverwood High School was asked to draw a model of a mixture composed of an element and a compound.
Comment on the usefulness of the students drawing.
Answer # 6
Sample Problem # 7
Finish Up Sample Problem # 7 in your group.
Remember to turn the packet in before you leave.
If you didn’t finish it then it is homework.