Balancing Chemical Equations Worksheet
advertisement

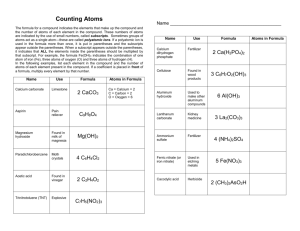
BALANCING CHEMICAL EQUATIONS Subscript – a number that goes on the bottom of an atom that tells us the atoms are glued together. You CANNOT change a subscript in balancing a chemical equation COEFFICIENT - a number that goes in front of an atom or compound that tells us how many of them we have So far, we have been doing simple reactions – adding one element or compound to another to make something new…These are called synthesis reaction It is now time to move on toe balanced reactions and balanced equations that more correctly show what actually happens in a chemical reaction. LAW OF CONSERVATION OF MASS This law says that the total amount of atoms must always be the same before and after a Chemical reaction because…ATOMS CANNOT BE CREATED OR DESTROUYED UNDER ORDINARY CIRCUMSTANCES The Steps to use in balancing a chemical reaction Step 1 Check the valences to be sure the compounds are correct and fix any diatomic gasses – necessary. (Remember! Gases do NOT travel alone ) Step 2 Count all the atoms of the reactants and products Step 3 Pick an element that is NOT equal and add a COEFFICIENT Step 4 Continue adjusting coefficients until it all balances – same number of atoms on each side of the equation (reactants and products) Note * If a compound starts with “H” (hydrogen) it is probably an ACID NEVER change SUB-SCRIPTS! Adjust/balance an equation by inserting COEFFICIENTS NOTE:** To balance an equation, count the atoms on each side of the equation (reactant side and product side). They must be the same on each side. If they are not, use Coefficients ONLY to balance the number of atoms on each side…remember!! CANNOT change subscripts! Kinds of Reactions Synthesis Reactions - 2 or more single elements come together to form a compound Example H2 + O2 2H2 + O2 -------> -----_> H2 O 2H2O Single Displacement Reactions - one metal knocks another metal out of a compound and takes its place Example Na + H2O -------> 2Na + 2H2O ---- NaOH + H 2NaOH + H2 Double Displacement Reaction - 2 metals switch places Example FeCl + NH4OH --- Fe(OH) + NH4Cl 2FeCl3 + 3NH4OH ---- Fe(OH)3 + 3NH4Cl Decomposition Reaction - A compound gets broken down into simpler parts Example KClO3 --- KCl + O2 2KClO3 ----> NOTE!** 2KCl + 3O2 To fully understand how to balance chemical equations YOU MUST PRACTICE!! See me for individual instruction if you are not getting this!