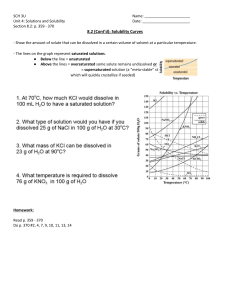
CHM01 – Chemistry for Engineers – Laboratory EXPERIMENT NO. 3 CONCENTRATION INTRODUCTION A solution may be classified whether dilute or concentrated. This is qualitatively described based on the relative amount of solute against solvent. Quite differently, a solution may be classified as unsaturated, saturated, or supersaturated. Their difference lies on the concentration relative to the saturation value at a particular temperature. There are several ways to express concentration. The simplest method is the percent by mass and percent by volume in which the mass or volume of the solute is divided by the total mass or total volume of the solution and multiplies by a hundred percent. This is frequently used in laboratories not only in schools but also in some industries. Molarity, which is the number of moles in a liter of the solution is equally useful in pharmaceutical industry in particular and chemical industry in general. Substances have different solubility in a particular solvent. Compounds which are less soluble produce a dilute solution yet a saturated one. Conversely , a compound may produce a concentrated and at the same time saturated if it has a relatively high solubility. OBJECTIVES 1. To understand how the concentration changes as more solute is being added. 2. To appreciate the concept of solubility and saturation in solutions. PROCEDURE 1. Access this link as the simulator that will be used in this activity: https://phet.colorado.edu/sims/html/concentration/latest/concentration_en.htm l 2. Set the solvent at 0.5 L. Change the solute samples once you determined the amount needed for the solution to be saturated. Use the concentration detector to determine the required amount. QUESTIONS 1. Know the saturation value of the substances used in the simulation. Which one is the most soluble (in g/L)? Which has the highest molarity at its saturation point? Encircle your answer in the table. Solute cobalt (II) nitrate cobalt (II) chloride potassium dichromate potassium chromate nickel (II) chloride copper (II) sulfate potassium permanganate sodium chloride Chemical Formula Co(NO3)2 Saturation Value (g/L) 1031 CoCl2 562.2 K2Cr2O7 150.0 K₂CrO₄ 650.5 NiCl₂ 675.2 CuSO4 220.2 KMnO₄ 75.9 NaCl 359.4 2. What is the solvent temperature used for this simulation? Refer to the Solubility curve on the last page. Solute potassium dichromate sodium chloride Temperature ( °C) 28 30 APPLICATION PROBLEMS. Follow strictly the rules on Significant Figures.