Acids and Bases in Aqueous Solution Worksheet
advertisement

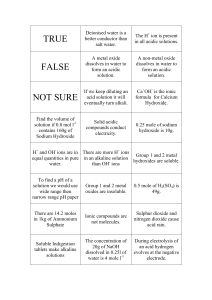
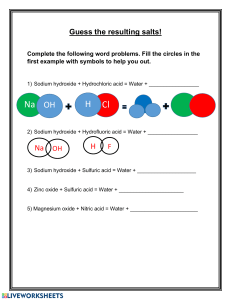
Acids and Bases in Aqueous Solution Acid-Base Theory An acid is a proton donor and a base is a proton acceptor (a) acids as proton donating species (molecule or ion) e.g. sulfuric acid H2SO4 H+ + _______________ b) bases as proton accepting species (molecule or ion) e.g. ammonia NH3 NH3 + H+ _________ (c) amphiprotic species i.e. a molecule or ion which can act as an acid or a base hydrogen carbonate ion HCO3- HCO3– + H+ _________ HCO3– H+ + _________ (d) conjugate pairs When a species behaves as an acid it donates a proton and forms a conjugate base When a species behaves as a base it accepts a proton and forms a conjugate acid eg: NH3 + ____________ H2O NH4+ + ____________ ______________ OH – _______________ An acid and its conjugate base differ by a PROTON (hydrogen ion) Compare the strength of H2SO4 and HSO4- Strength of Acids and Bases Strengths of acids may be compared by the extent of their proton transfer to water forming aqueous hydronium ions. In other words, a strong acid ionises to a very great extent. strong acid: HCl + H2O ___________ + ___________ practically all HCl molecules have ionised weak acid: CH3COOH + H2O ___________ + ___________ few CH3COOH molecules have ionised. Examples of strong acids: ______________________________________________ Examples of weak acids: _______________________________________________ The strengths of bases may be compared by the extent to which they produce OHions in solution. 1. Strong base: NaOH _________ + _________ This ionisation proceeds to a very great extent 2. Weak base: NH3 + H2O ________ + ________ This ionisation proceeds to a very limited extent. Examples of Strong Bases: ___________________________________________ Examples of Weak Bases: ____________________________________________ Acid-Base nature of Salts A salt can be acidic, basic or neutral depending how the cation or anion present reacts with water. For example, ammonium (NH4+) salts tend to be acidic, because the NH4+ ion is the conjugate acid of a weak base, ie: it is a strong acid. This can be shown by the following hydrolysis reaction: NH4+ Cl– +H2O NH3 + H+ happens to a _________ extent HCl + OH – happens to a ____________ extent So ammonium chloride is an acidic salt, because the ammonium ion came form a weak base (NH3) and the chloride came from a strong acid (HCl). Sodium ethanoate is a basic salt because sodium came from a strong base (NaOH) and ethanoate came from a weak acid (CH3COOH) Worked example 14 All of the hydrogen halides are acidic in aqueous solution. Explain any trend in acid strength related to the halogen's position in the group. Kw and pH Calculations Water ionises to H+ ions and OH- ions to a very limited extent. H2O H+ + OH – The equilibrium constant for this ionisation is very small at 10-14 (at 25C) i.e. K w 10 14 [ H ][OH ] 1 [a useful extension of this is pKw = 14 = pOH + pH] Because water is neutral, [H+] = [OH-] = ________ pH log[ H ] Worked example 15 Find the pH of a 0.001 molL-1 solution of Ca(OH)2 solution. Amphoteric Substances An amphoteric substance reacts with both acids and bases. Chromium, zinc and aluminium are amphoteric metals and they form amphoteric oxides and amphoteric hydroxides. These metals form complex ions with bases. eg, Aluminium: Al(s) + 2OH –(aq) + 6H2O 2Al(s) + 6H+(aq) 2[Al(OH)4] –(aq) + 3H2(g) 2Al3+(aq) + 3H2(g) Complex Ions A complex ion consists of a positive ion (usually a transition metal ion) surrounded by a number of ligands (ions or polar molecules). Eg: [Fe(OH)6]3+, [Al(OH)4]3+ , [Cu(NH3)4]2+ Cu2+ Volumetric Analysis You must know the steps that need to be followed when carrying out volumetric analysis - they are not presented here, but are available in your text book and laboratory manual. Be able to describe (i) how to make a primary standard, (ii) do an accurate titration, (iii) explain these curves and (iv) discuss the sources of error. pH 14 pH 14 0 0 Volume of acid added Volume of acid added 14 pH pH 14 0 0 Volume of acid added Volume of acid added Indicators: Indicator Methyl Orange Phenolphthalein Useful pH range 3.1 – 4.4 8.3 - 10 Colour Change red - yellow colourless - red (pink) It is crucial that an indicator is chosen so that the end point of the titration corresponds to the equivalence point of the neutralisation. Acid-Base Calculations Stoichiometry is applied in acid-base calculations in exactly the same way as it is applied in any other area of Chemistry. The four steps of stoichiometry apply (see earlier) Worked example 16: Barium hydroxide is normally hydrated, this means it is of the form: Ba(OH)2.xH2O. 0.816 g of a sample of hydrated barium hydroxide was dissolved in 25.0 mL of water and titrated against 0.177 molL-1 hydrochloric acid. The titration required exactly 29.3 mL of the acid for complete neutralisation. (a) State a suitable indicator for the titration. Explain your choice. (b) Find the correct formula for the Ba(OH)2.xH2O. Practice Questions 1. Ammonium nitrate solution is (a) (b) (c) (d) (e) 2. Which of the following solutions has the lowest pH? (a) (b) (c) (d) 3. 0.1 M ethanoic acid 0.1 M hydrochloric acid 0.2 M sodium hydroxide 0.2 M nitric acid. Which of the following descriptions best describes 10 mol L-1 ammonia? (a) (b) (c) (d) 4. Acidic because nitrate ions react with water to give HNO3 Basic because ammonium ions react with water to give OHNeutral because ammonia is basic and nitric acid is acidic. Acidic because ammonia reacts with water liberating H3O+ ions. Basic because it contains ammonia which is a base. a dilute solution of a weak base a concentrated solution of a weak base a dilute solution of a strong base a strong solution of a weak base Which of the following has a pH less than 7? (a) (b) (c) (d) 0.01 mol L-1 ammonium chloride 0.01 mol L-1 calcium hydroxide 0.01 mol L-1 potassium nitrate 0.01 mol L-1 sodium ethanoate 6. Ethanoic acid is titrated against sodium hydroxide (in the burette), using methyl orange as the indicator. Which of the following statements about the titration is true? (a) (b) (c) (d) 7. The end point and equivalence point occur at the same time. The end point occurs after the equivalence point. The end point occurs before the equivalence point. No end point would occur. Classify the following solutions as acidic, basic or neutral. Where the solution is basic or acidic, write a hydrolysis equation to support your answer. KH2PO4 8. MgF LiHCO3 KCl Chemists can standardise sodium hydroxide solutions with the monprotic acid potassium hydrogen phthalate (KHC8H4O4) as a primary standard. A 25.0 mL aliquot of a sodium hydroxide solution requires 23.1 mL of 0.0994 mol L-1 potassium hydrogen phthalate for complete reaction. (a) Suggest an indicator for the titration, explain your choice. (b) Explain what is meant by a primary standard. (c) Why do you think the chemist standardised the sodium hydroxide solution? (d) 9. Find the concentration of the sodium hydroxide solution. Orange juice contains the weak acid citric acid. The formula is shown in the figure.: O H OH H O C C C C C HO H C H HO O (a) State what is meant by the term weak acid. (b) Each molecule of citric acid contains 8 hydrogen atoms. Only three of these can be considered as acidic hydrogens. Identify these by circling them on the diagram. (c) 24.2 g of a particular brand of orange juice was placed in a conical flask. Titration with sodium hydroxide solution showed that the orange juice contained 0.700 % citric acid by mass. (d) (i) Calculate the mass, and hence the number of moles of citric acid in the sample of orange juice. (ii) How many moles of sodium hydroxide were required to react completely with this amount of citric acid. OH Exemplar exam (Section 1) Question 15 Which of the following solutions can be added to 100.0 mL of a 0.2 mol L-1 NH3 solution to produce a buffer? I 100.0 mL of 0.20 mol L-1 NH4Cl(aq) II 50 mL of 0.20 mol L-1 HCl(aq) III 100.0 mL of 0.20 mol L-1 HCl(aq) a) b) c) d) I only I and II only II and III only I, II and III Question 16 Pure water undergoes self-ionisation according to the equation: 2H2O() H3O+(aq) + OH-(aq) The equilibrium constant for the reaction is: 1.0 x 10-14 at 25oC 5.5 x 10-13 at 100oC Which one of the following statements is correct? a) At 100oC the pH of water is less than 7, but the water is still neutral. b) At 100oC the pH of water is less than 7, and therefore the water is acidic. c) At 100oC the pH of pure water is greater than 7.0, and therefore the water is basic. d) At 100oC the pH of water is greater than 7, and therefore the water is acidic. Question 17 Potassium permanganate solution is standardised using the following procedure: sulfuric acid is added to 25.00 mL of a standard solution of oxalic acid (ethandioic acid) and the mixture warmed and titrated with potassium permanganate solution. Which one of the following will cause an error? a) Leaving the conical flask (titration vessel) wet after rinsing with distilled water. b) Rinsing the burette with distilled water, then with a little of the permanganate solution, and then filling it with the permanganate solution. c) Rinsing the pipette with distilled water, then using it to dispense the oxalic acid solution. d) Washing down the sides of the conical flask with distilled water from time to time during the titration. Buffers Exemplar from (Section 2) [9 marks] The pH of blood is maintained through buffering. The major buffer system present in blood is based on a carbonic acid/hydrogencarbonate ion buffer. When carbon dioxide enters the blood stream, the following reaction occurs: CO2(g) + 2 H2O() H2 CO3( aq ) + H 2 O ( ) + H3 O(aq) HCO 3(aq) The presence of carbonic acid and hydrogencarbonate ions maintains the pH of blood to about 7.4. (a) Excess HCO 3(aq) produced as a result of strenuous exercise is removed from the blood by the kidneys. (i) Describe the immediate change in the pH of the blood. [1 mark] __________________________________________________________ __________________________________________________________ (ii) Explain the immediate change in the pH. [2 marks] _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ (b) Hyperventilation, or rapid breathing, decreases the amount of carbon dioxide in the lungs. As a consequence the concentration of carbon dioxide dissolved in the blood also decreases. (i) Describe the immediate change in the pH of the blood. [1 mark] __________________________________________________________ __________________________________________________________ __________________________________________________________ __________________________________________________________ (ii) Explain the immediate change in the pH. [2 marks] _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ (iii) Describe how the buffer system counteracts this change in pH. [3 marks] _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________