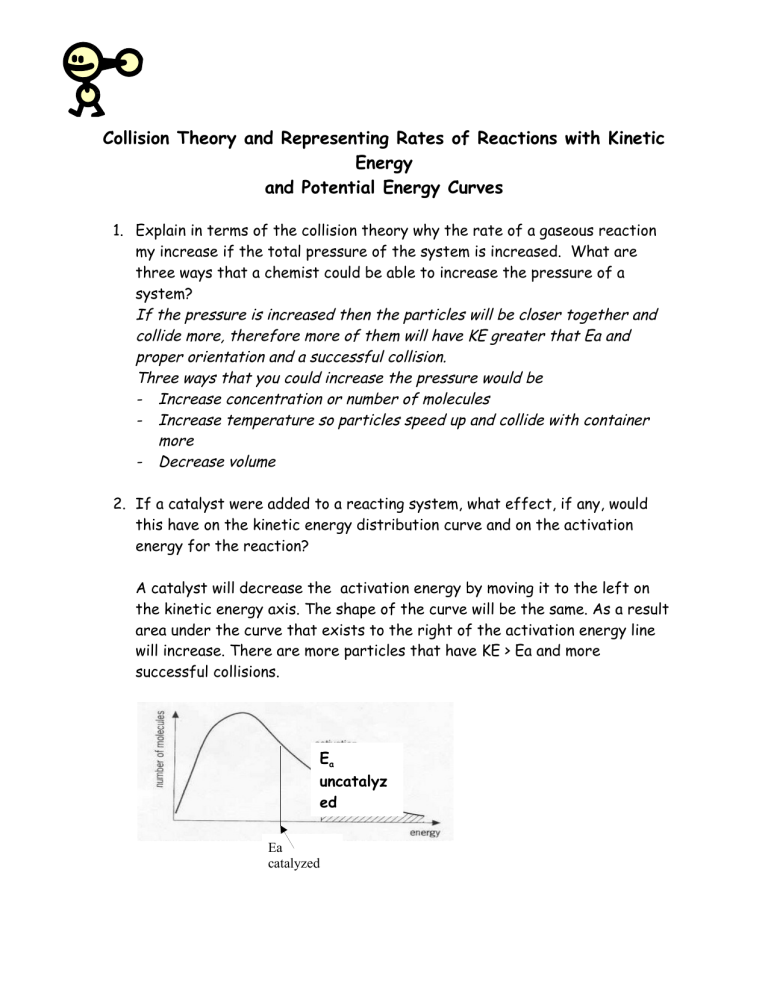
Collision Theory and Representing Rates of Reactions with Kinetic Energy and Potential Energy Curves 1. Explain in terms of the collision theory why the rate of a gaseous reaction my increase if the total pressure of the system is increased. What are three ways that a chemist could be able to increase the pressure of a system? If the pressure is increased then the particles will be closer together and collide more, therefore more of them will have KE greater that Ea and proper orientation and a successful collision. Three ways that you could increase the pressure would be - Increase concentration or number of molecules - Increase temperature so particles speed up and collide with container more - Decrease volume 2. If a catalyst were added to a reacting system, what effect, if any, would this have on the kinetic energy distribution curve and on the activation energy for the reaction? A catalyst will decrease the activation energy by moving it to the left on the kinetic energy axis. The shape of the curve will be the same. As a result area under the curve that exists to the right of the activation energy line will increase. There are more particles that have KE > Ea and more successful collisions. Ea uncatalyz ed Ea catalyzed 3. A)Explain in terms of collision theory why a log burns slowly in air while if sawdust made from the same log was scattered throughout the room and ignited it burns explosively. The sawdust particles have more surface area to react with the oxygen and flame. ( more collisions with oxygen molecules per unit time).This will allow for more potentially successful collisions leading to more product. b) Construct a labelled kinetic energy diagram to represent both situations. Draw curve. Ea stays in the same position. More area will be to the right of the Ea in your graph. Make sure axis is labelled with KE and fraction of collisions . 4. For the reaction CO + NO2 CO2 + NO , the activation energy for the forward reaction is known to be 134 kJ. a) Use the Standard molar enthalpies table ( pg 597) and Hess’ Law to find the Enthalpy of this reaction. ∆Hrxn = ∑∆Hprod - ∑∆Hreact my table may be different but you get idea = [∆H CO2 + ∆H NO] – [∆H CO + ∆H NO2] = [40.37 – 393.5] – [ -110.5 + 33.8] = -303.13 + 76.7 = - 226.4 kJ exothermic b) Using both of these values, construct a labelled potential energy diagram for the reaction. - Are axis labelled PE and Rxn Coordinate - Are products lower than reactants and the difference between the two labelled as ∆H= - 226.4 kJ - Ea f was given . Is it written on graph - Ea r is sum of 226.4 and 134. Is it written on graph as 360.4 kJ?