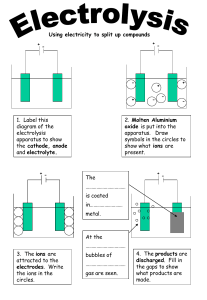
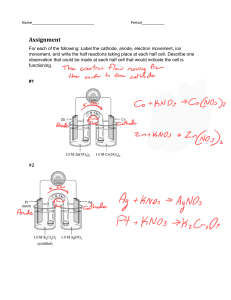
1 ELECTROLYSIS 01. Definitions: i) Electrolysis: The decomposition of an ionic compound either in molten state or in aqueous state into its constituents after passing electric current through it is known as electrolysis. ii) Electrolyte: An electrolyte is a compound in solution or a molten compound which conducts electric current with the decomposition at the electrodes. iii) Non- electrolyte: A non-electrolyte is a solution or a molten compound which cannot be decomposed by an electric current. iv) Strong electrolyte: An electrolyte which ionizes completely. v) Weak electrolyte: An electrolyte which ionizes partially. vi) Electrodes: The electrodes are two poles of carbon or metal at which the current (as a flow of electrons) enters and leaves an electrolyte. vii) Anode: The electrode which is attached to the positive end of the battery. viii) Cathode: The electrode which is attached to the negative end of the battery. 2 ix) Anion: Negatively charged ion. During electrolysis, it moves to the anode. x) Cation: Positively charged ion. During electrolysis, it moves to the cathode. xi) Inert electrode: Do not take part in any chemical reaction during electrolysis. Example: graphite (carbon) and platinum. xii) Reactive electrode: Take part in chemical reaction during electrolysis. Example: all electrodes except graphite and platinum. xiii) Oxidation: Loss of electron by a substance. During electrolysis oxidation always takes place at anode. xiv) Reduction: Gain of electron by a substance. During electrolysis reduction always takes place at cathode. xv) Discharge: The process of gaining or losing electrons at the electrodes is known as discharge. When ions are discharged at the electrodes, they form atoms or molecules. xvi) Binary compound: A compound containing only two elements. A binary compound often contains a metal cation and a non-metal anion. 3 02. When electricity is passed through an electrolyte: When electricity is passed through an electrolyte, chemical reactions take place at the electrodes and the electrolyte is decomposed. The reactions taking place at the electrodes are called electrolytic reactions. 03. Flow of electrons: Electrons the enter external circuit through cathode and leaves the external circuit through anode. 04. Existence of mobile ions: Electrolytes only conduct electricity because they contain mobile ions. Molten or aqueous ionic compounds can acts as an electrolyte, so they must contain mobile ions. In the ionic compounds, oppositely charged ions are held fixed in a lattice in solid state. Ions become immobile. They are unable to move freely thus unable to conduct electricity. 4 05. Differences between electrical conductor and electrolytic conductor: Metals, carbon (graphite) and ionic compounds (molten or aqueous) are conductors of electricity. Both metals and graphite are known as electrical conductor and ionic compounds are known as electrolytic conductor. 06. Electrical Electrolytic conduction conduction Electricity is Electricity is conducted by the conducted by the Method of flow of electrons movement of positive conduction from one end of the ions and negative conductor to the ions across the other end. electrolyte. Metals and graphite The electrolytes Effect of remain unchanged are decomposed to conduction chemically when an form new substances electric current when they conduct flows through them. electricity. Electrolysis of water: Pure water is a poor conductor of electricity because it consists almost entirely of molecules and has very few ions in it. To increase the conductivity of pure water, small amount of concentrated sulfuric acid or an ionic compound is to be added. The products of electrolysis of water are always two volumes of hydrogen at the cathode and one volume of oxygen at the anode. 5 07. Electrolysis of molten lead (II) bromide [PbBr2] using inert electrodes. Ions present: Pb2+ and BrAt the anode: 2Br-(l) → Br2 (l) + 2eAt the cathode: Pb2+(l) + 2e- → Pb(s) Observations: 08. i) Red brown vapour around anode. ii) Solid deposited at the bottom of the cathode. Selective discharge of ions: i) Position of the metal or group in the electrochemical series: Less reactive ion will discharge. More reactive ion will remain in the electrolyte. [This factor is applicable to both anion and cation.] ii) Concentration: If reactive anion has greater concentration it will discharge. [This factor is applicable to anion only.] iii) Nature of the electrode: Use of reactive electrode instead of inert electrode, influence the choice of ion discharge. In the electrolysis of copper (II) sulfate solution using copper electrodes, the anode dissolves. Thus, produces more copper (II) ions in the electrolyte. 6 09. Reactivity of anions: Left to right reactivity decreases. SO42-, NO3-, Cl-, Br-, I-, OH- 10. Reactivity of metals: Left to right reactivity decreases. PoSoCaMAl (C) ZIL (Hy) CoMSG 11. Electrolysis of dilute sulfuric acid [H2SO4] using inert electrodes. Ions present: H+ , SO42- , OHAt the anode: 4OH-(aq) → 2H2O(l) + O2(g) + 4eAt the cathode: 4H+(aq) + 4e- → 2H2(g) [Ultimately electrolysis of water.] 12. Electrolysis of copper (II) sulfate [CuSO4] solution using inert electrodes. Ions present: Cu2+ , SO42- , H+ and OHAt the anode: 4OH-(aq) → 2H2O(l) + O2(g) + 4eAt the cathode: 2Cu2+ (aq) + 4e- → 2Cu(s) Observations: i) Blue colour of the solution turns colourless. ii) Cathode gets thicker. 7 13. Electrolysis of copper (II) sulfate [CuSO4] solution using copper electrodes. Ions present: Cu2+ , SO42- , H+ and OHAt the anode: Cu(s) → Cu2+ (aq) + 2eAt the cathode: Cu2+ (aq) + 2e- → Cu(s) Observations: iii) Blue colour of the solution remain unchanged. iv) Cathode gets thicker. ∗ Important information: Impure copper is purified by this method. Impure copper is made anode and pure copper is made cathode. Copper purified by this method is 99.999% pure. Impurities include platinum, silver and gold. These metals are deposited at the bottom of the cathode. These metals are separated and sold out. Deposited impurities are collectively known as sludge. 14. Electrolysis of dilute sodium chloride [NaCl] solution using inert electrodes. Ions present: Na+ , Cl- , H+ and OHAt the anode: 4OH-(aq) → 2H2O(l) + O2(g) + 4eAt the cathode: 4H+(aq) + 4e- → 2H2(g) [Ultimately electrolysis of water.] 8 15. Electrolysis of concentrated sodium chloride [NaCl] solution (Brine) using inert electrodes. Ions present: Na+ , Cl- , H+ and OHAt the anode: 2Cl-(aq) →Cl2(g) + 2eAt the cathode: 2H+(aq) + 2e- → H2(g) ∗ Important information: Ions left behind are Na+ and OH- . Sodium hydroxide is manufactured by this method. 16. Electroplating: The process in which a layer of a metal is deposited on metallic or non-metallic electrode by electrolysis in an electrolytic cell is known as electroplating. 9 a) b) 17. Conditions for electroplating: i) Object is made cathode ii) Anode is the electroplating metal iii) Electrolyte is the electroplating metal ion solution. Uses: i) In making inexpensive jewelry. ii) For decorative purposes. iii) Increases the life of the metal. iv) Prevents corrosion. v) In industries. Copper plating: The anode is pure copper, often called plating copper. The metal object to be copper-plated is made the cathode and the electrolyte is copper (II) sulfate solution. At the anode: Cu(s) → Cu2+ (aq) + 2eAt the cathode: Cu2+ (aq) + 2e- → Cu(s) 10 18. Simple cells: A simple cell is a device that converts chemical energy into electrical energy. A simple cell consists of two electrodes (two different metals) and an electrolyte solution (acid, alkali or salt solution). a) How simple electric cell work: The working principle of simple electric cell depends upon the principle that, whenever two dissimilar metals are immersed inside an electrolyte solution, the more reactive metal will have a tendency to dissolve in the electrolyte as positive metal ions, leaving electrons behind on the metal plate. This phenomenon makes the more reactive metal plate negatively charged. The cell voltage depends on the positions of the metals in the reactivity series. The further apart the metals are in the reactivity series, the bigger the voltage of the cell. 11 19. Extraction of aluminium by the electrolysis of purified aluminium oxide [Al2O3] / alumina. The steel container is coated with graphite and this is used as the cathode. The aluminium ore bauxite [Al2O3, 2H2O] is purified to yield aluminium oxide, alumina. To the molten cryolite in the steel tank, alumina is added. Blocks of graphite are dipped into the electrolyte solution which is used as anode. At the anode: 6O2-(l) → 3O2(g) + 12eAt the cathode: 4Al3+(l) + 12e- → 4Al(l) Molten aluminium runs down to the bottom of the cell where it can be trapped off as pure liquid aluminium. The oxygen formed at the anode reacts with carbon (graphite) to form carbon dioxide. C(s) + O2(g) → CO2(g) Thus, the graphite anode is eaten away by hot oxygen and needs to be replaced frequently. 12 Important information: The overall reaction is 2 Al2O3(l) → 4Al(l) + 3 O2(g) Cryolite is used to reduce the melting point of alumina. Melting point of alumina, Al2O3 is 20500 C. a) Cryolite: i) Formula: Na3AlF6 ii) Systemic name: Sodium hexafluoroaluminate (III) Properties of aluminium: i) It is a strong, malleable and ductile metal element. i) It has a low density. ii) It is resistant to corrosion. iii) It is a good conductor of heat and electricity. iv) It can be polished to give a highly reflective surface. v) It is durable. vi) It is odorless. vii) It is impermeable. viii) It is 100% recyclable with no deterioration in quality. ix) It is non-toxic. b) The apparent lack of reactivity of aluminium: Aluminium forms a protective oxide layer which makes it appear to be less reactive than it really is. When this layer is removed, the observations are more reliable. 13 c) Uses of aluminium: Sl. No. 01 Use of aluminium Aircrafts Reason Light, strong, corrosion resistant 02 Other transports, such as Light, strong, corrosion ships superstructures, resistant container vehicle bodies and metro trains 03 Overhead power cables (with Light, strong, corrosion a steel core to strengthen resistant, good conductor of them) electricity Light, strong, corrosion 04 Cookers, cook wares, boilers resistant, good appearance, good conductor of heat. Mirrors, reflectors and heat 05 High reflectivity resistant clothing for fire fighting. 06 Drink cans, roofing materials Light, strong, corrosion resistant 07 Window frames, greenhouses Corrosion resistant, low density 08 Food container Light, strong, corrosion resistant 14 d) Recycling aluminium: Advantages: i) Saves a lot of energy. ii) Mining avoided iii) It never wears out iv) Reduces the emission of greenhouse gases v) Reduces the use of natural resources and chemicals vi) Sustainability increases jobs. Disadvantages: i) Need to be separated from plastic, steel and debris. ii) Sorting is time consuming and costs money iii) High energy cost in reprocessing and transporting materials iv) When aluminum is continually recycled it loses it quality. So, a product made with new aluminum will be of higher quality than the recycled product.