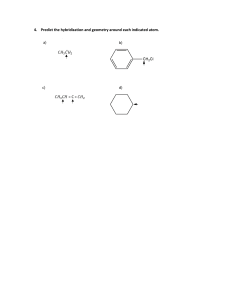
Lecture 7 CHEM101 Hybridisation of Atomic Orbitals Dr. Noha Osman Learning Outcomes • • • • Understand the valence bond theory Understand the concept of hybridization. Understand the different types of orbital hybridization. Understand the relationship between hybridization and molecular geometry • Determine the effect of multiple bonds. • Be able to predict the type of hybridization in given covalent compounds. • Know the different types of intermolecular forces. 2 Covalent Bonding • Lewis theory: covalent bond is merely formed by the sharing of electrons. H-H ≠ F-F (different bond enthalpies and bond lengths) • Quantum mechanics: more complete explanation of covalent bonding; two theories were postulated: 1- Valence Bond Theory: Uses atomic orbitals of individual atoms to explain covalent bonding. 2- Molecular Orbital Theory: Assumes the formation of molecular orbitals to explain covalent bonding. 3 Valence bond theory This theory assumes that a covalent bond is formed when: 1- atomic orbital of one atom merges with that of another atom. The orbitals are then said to share a common region of space, or to overlap. 2- Overlap results in energy release, therefore the energy of the system reaches a minimum and is most stable (bond formation: exothermic reaction!) 4 Hybridisation • VB theory uses hypothetical hybrid orbitals, which are atomic orbitals obtained when two or more nonequivalent orbitals of the same atom combine in preparation for covalent bond formation. • Hybridization is the term applied to the mixing of atomic orbitals in an atom (usually a central atom) to generate a set of equivalent hybrid orbitals. • The valence orbitals, associated with the highest principal quantum level that contain electrons on a given atom are responsible for hybridization. 5 Real Life Hybridisation A ZONKEY is a HYBRID of a DONKEY and a ZEBRA !!! 6 Types of Hybridisation sp3 Hybridisation sp2 Hybridisation sp Hybridisation sp3d Hybridisation sp3d2 Hybridisation 7 sp3 Hybridisation, CH4 molecule The electronic configuration of C is 1s2 2s2 ↑↓ 2p2 ↑ ↑ • It might be expected that C would form only two bonds with 2 H atoms, since it has two unpaired electrons. • However, it actually forms four C-H bonds in methane! • This can be explained by assuming that an electron from the 2s is energetically excited to the empty 2p orbital: 2p3 2s1 ↑ ↑ ↑ ↑ Thus, four bonds can be formed: 1 bond: overlap of 2s orbital of C and 1s orbital of H 3 bonds: overlap of 2p orbitals of C with 1s orbitals of three H atoms. 8 sp3 Hybridisation, CH4 molecule (continue) • Since all four C-H bonds are identical, then this means that all four C orbitals involved in bonding are identical, too! • This can be explained by: Hybridisation 2p sp3 sp3 C-H bonds 2s 1s Electron Promotion 1s Hybrid Orbitals 1s Forming Bonds 9 sp3 Hybridisation Four hybrid orbitals are directed toward the four corners of a regular tetrahedral. CH4 has a tetrahedral shape, and all the HCH angles are 109.5°. 10 sp3 Hybridisation, NH3 molecule 2p sp3 lone pair 2s 1s N atom (ground state) sp3 1s N atom (hybridized state) N-H bonds 1s N atom (in NH3) 11 sp3 Hybridisation, NH3 molecule (continue) Three of the four hybrid orbitals form covalent N-H bonds The fourth hybrid orbital accommodates the lone pair on nitrogen Repulsion between the lone-pair electrons and bonding-electron pairs decreases the HNH bond angles from 109.5° to 107.3° Thus, Hybridisation and VSEPR model are RELATED! 12 How to determine the type of hybridization? Count the number of effective electron pairs around the atom. What is considered as an effective electron pair? Lone pair. Single bond. Double bond. Triple bond. Each of the above is counted as one effective electron pair. 13 Relationship between the number of effective pairs and their hybrid orbitals No of effective electron pairs Type of hybridization Arrangement Of hybrid orbitals 2 sp Linear 3 sp2 Triangular planar 4 sp3 Tetrahedral 5 sp3d Triangular bipyramidal 6 sp3d2 Octahedral 14 sp2 Hybridisation, BF3 molecule B: ground state electron configuration, can form only one bond! B: energy promoted an electron from the 2s to an empty 2p orbital, thus can form three bonds! Since all B-F bonds are identical, therefore sp2 hybridization must have occurred! sp2 orbitals Empty p orbital 15 sp2 Hybridisation, BF3 molecule (continue) Three hybrid orbitals are directed toward the 3 corners of an equatorial, planar triangle. BF3 has a trigonal planar shape, and all the FBF angles are 120°. 16 Types of Covalent Bonds VB Theory: Two ways orbitals can overlap to form covalent bonds between atoms Sigma Bonds () - end-to-end orbital overlap. - electron density concentrated between the nuclei of the bonding atoms. Pi Bonds () - Sideways orbital overlap. - electron density concentrated above and below the plane of the nuclei of the bonding atoms. 17 sp2 Hybridisation, C2H4 molecule C: ground state electron configuration. C: energy promoted an electron from the 2s to an empty 2p orbital. sp2 hybridised state sp2 orbitals 2p orbital 18 sp2 Hybridisation, C2H4 molecule (continue) The three sp2 orbitals on each carbon atom form two bonds with the two hydrogen 1s orbitals and a third bond with the sp2 hybrid orbital of the adjacent C atom. The two unhybridised 2p orbitals of each of the C atoms form a bond by overlapping sideways. 19 sp Hybridisation, BeH2 molecule Be: ground state electron configuration, cannot form any bonds! Be: energy promoted an electron from the 2s to the 2p orbital, thus can form two bonds! Since both Be-H bonds are identical, therefore sp hybridization must have occurred! sp Hybridisation, BeH2 molecule (continue) Be atom: 1s atomic orbital of H .. .. 1s atomic orbital of H Overlap region 21 sp Hybridisation, CO2 molecule Carbon atom sp orbitals Two 2p orbitals Two effective pairs; sp hybridization .. .. O=C=O .. .. Oxygen atom sp2 orbitals 2p orbital Three effective pairs; sp2 hybridization 22 sp Hybridisation, CO2molecule (continue) Note that two bonds are formed between the two sp hybridized orbitals of C and sp2 hybridized orbitals of each of the O atoms. Note that the two 2p orbitals remain unchanged on the sp hybridized carbon. These are used to form the two bonds with the 2p orbitals on each of the oxygen atoms. 23 sp Hybridisation, N3 molecule :NN: sp orbitals Two 2p orbitals Two effective pairs; sp hybridization 24 sp3d Hybridization • E.g. SF4 & PCl5 : a set of five effective pairs around the S P P atom is available. • In general, a set of five effective pairs around a given atom always requires a trigonal bipyramidal arrangement, which in turn requires sp3d hybridization of that atom. sp3d2 Hybridization • • E.g. SF6 (6 bonding pairs) & XeF4 (4 bondig pairs + 2 lone pairs around Xe). In general, six electron pairs around an atom are always arranged octahedrally and require sp3d2 hybridization of the atom. 25 Hybridization with d-Orbitals, 3rd Row: Extension of Octet Rule s p d Hybridization F F sp3d S: 3s23p4 F F F Bonding F lone pair trigonal-bipyramidal SF4 S F F s p d Hybridization Bonding F F F F F F S: 3s23p4 sp3d2 F octahedral F SF6 S F F F 26 F Practice Excercise For each of the following molecules or ions, predict the hybridization of each atom and describe the molecular structure. a. CO The Lewis structure is :C O: Each atom has two effective pairs, which means that both are sp hybridized. 27 b. BF4The Lewis structure for such ion is as above. Four pairs of electrons around B, thus, sp3 hybridized; tetrahedral structure. 28 Intermolecular Forces Intramolecular or bonding forces are found within a molecule. Intermolecular or nonbonding forces are attractive forces found between molecules. Intermolecular forces are relatively weak compared to bonding forces and thus require less energy to overcome. They are responsible for physical properties of matter. 29 Types of Intermolecular Forces van der Waals Ion-Dipole Forces Hydrogen Bonding 30 DipoleDipole Forces Ioninduced dipole forces van der Waals Dipoleinduced dipole Forces London Dispersion forces 31 van der Waals 1- Dipole-Dipole Forces • Are attractive forces found between polar molecules, that is, molecules that possess net dipole moments. • Molecules align themselves to maximize attractive interactions while minimize repulsive interactions. • Dipole forces are 1% as strong as covalent bonds and become weaker especially in the gas phase where the distance between dipole increases. The positive pole of one polar molecule attracts the negative pole of another. solid 32 2- Ion-induced dipole interaction Are attractive forces that arise between ions and temporary dipoles induced in atoms or molecules. 3- Dipole-induced dipole interaction Are attractive forces that arise between dipoles and temporary dipoles induced in atoms or molecules. 33 4- London Dispersion forces • Non-polar molecules and noble gases exhibit temporary dipoles, where it happens that for a very short time period the electrons are not distributed symmetrically around the molecule (the electron cloud is more dense on one side of the molecule in comparison to the other) leading to temporary δ+ and δ- charges. (Instantaneous dipole) • This instantaneous dipole distorts the electron distribution in neighboring molecules thus inducing dipoles in these neighboring molecules. (Induced dipole) • This leads to interatomic or intermolecular attraction that is relatively weak and short lived. 34 Ion-Dipole forces • Are forces which attract an ion and a polar molecule to each other. • Strength of interaction depends on the charge and size of the ion and on the magnitude of the dipole moment and size of the molecule. Interaction of a water molecule with a Na+ ion and a Mg2+ ion. 35 Hydrogen Bonding • Hydrogen bonding is possible for molecules that have a hydrogen atom covalently bonded to a small, highly electronegative atom with lone electron pairs, specifically N, O, or F. • An intermolecular hydrogen bond is the attraction between the H atom of one molecule and a lone pair of the N, O, or F atom of another molecule. • ↑ ∆ EN → ↑ bond polarity → ↑ strength of H bond. 36 Comparison of Strengths of Intermolecular Forces Thank You