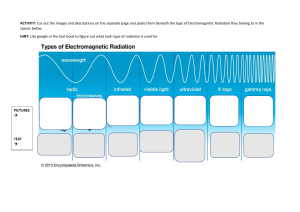
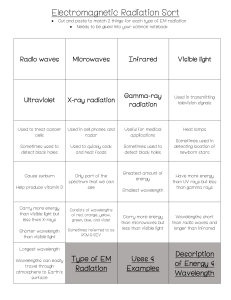
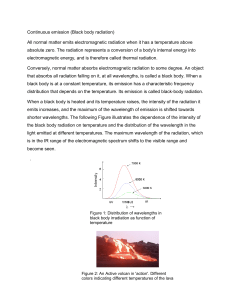
Problem Set 3.1 – Research Chemistry PLEASE work out on a separate paper and upload a picture. 1. What is the frequency (in Hertz) of electromagnetic radiation that has a wavelength of 1380 nm? 2. What is the wavelength (in Meters) of electromagnetic radiation that has a frequency of 5.4 x 1011 kHz? 3. What is the energy (in Joules) of the electromagnetic radiation in question 1? 4. A photon of light has a wavelength of 0.050 cm. Calculate its energy. Draw the electron configuration diagram for the following atoms and state how many valence electrons they have: 5. 6. 7. 8. 9. Beryllium Potassium Chromium Krypton Uranium Identify the following electron configuration diagrams: Nitrogen Oxygen Fluorine Neon