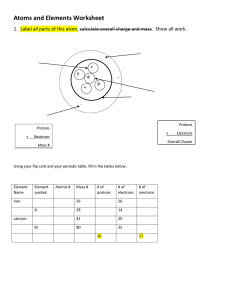
Atoms and Elements (LT 1-4) Essential Question- What is everything in the universe made of? What do you remember? Copying this slide is optional The periodic table shows approximately 120 known elements. Elements can be classified as metals, nonmetals, and metalloids. When two or more elements combine, they form a compound. Everything in the universe is made of elements which are made of atoms. Remember : Cornell Style Notes; On as many right hand pages as you need What are the elements made of? Element - the simplest pure substance Elements are made of atoms, and each element’s atoms are unique to that element. (ex: the element gold is made of gold atoms, the element oxygen is made of oxygen atoms, etc.) Atoms - the basic building blocks of all matter. They are the smallest part of an element that keeps that elements properties. 3 Subatomic particles : protons, neutrons, electrons What are the parts of an atom? (LT 1) 1. Nucleus –the small dense center of atom composed of protons and neutrons; this is where most of the atoms mass is located. (mass number if rounded. Atomic mass if not rounded) 2. Electron Cloud/Energy Levels – regions around the nucleus of an atom where electrons are located; mostly empty space Draw this image to help you remember. What are subatomic particles? (LT 2,3,4) a. Protons – positively charged particles inside the nucleus (center). b. Neutrons – neutral particles (have no charge)inside the nucleus. c. Electrons – negatively charged particles found in the electron cloud outside of the nucleus. Nucleus: protons +Neutrons = mass number Draw and label the atom above to help you remember. Make it large and neat. What is the mass of an atom? Atomic Mass Unit – (amu): Unit of measurement for the mass of subatomic particles. 1 amu = 1.7 X 10-24 g - They are TINY!!! (0.000000000000000000000017 grams) 1. Proton = 1 amu 2. Neutron = 1 amu 3. Electron = almost zero (TINIEST particle with the least mass) Most mass is in the nucleus with protons and neutrons *Otherwise atoms are mostly EMPTY SPACE 1. The atomic mass of an atom is the mass of the protons & neutrons added together. 2. When we round that number, it is called the mass number. Costa Questions and Summary Develop a Costa Question from your notes for each level. The 5-7 Sentence summary goes at the bottom of the page. It must answer the essential questions and describe each concept. Underline all of the vocabulary words in your summary.