Document 17623961

a nuclear reaction involves altering the number of protons and/or neutrons in an atom
release LOTS more energy than a chemical reaction
› uranium-235 can produce 3.7 million times the energy as an equal amount of coal
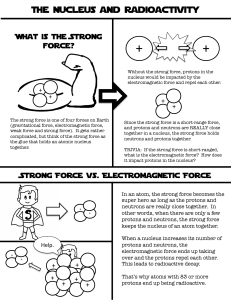
the nucleus of an atom becomes unstable if it contains too many, or too few neutrons, relative to the number of protons forces inside the atom result in it breaking apart or releasing particles
• nuclear fusion
the process of combining the nuclei (plural of nucleus) of lighter atoms to make heavier atoms
occurs in the Sun and the resulting energy released provides Earth with heat and light
nuclear fission is the process of splitting the nucleus of an atom
› only certain isotopes of certain elements are fissile (can split)
uranium is the principle element used in nuclear reactors and in certain types of atomic bombs
an unstable isotope will change into a more stable atom (even a different element)
› the nucleus spontaneously emits particles or energy as it stabilizes in alpha and beta decay, protons and neutrons are gained or lost creating a different element
in gamma decay, the nucleus gets rid of energy in the form of gamma rays, still the same element
Half-life
one half-life is the amount of time required for ½ of the original atoms in a radioctive sample to decay
Carbon dating (2:10)