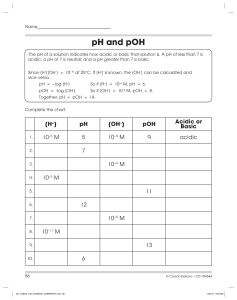
Name___________________ Period___________________ Chemistry Quiz
advertisement

Name___________________ Period___________________ Chemistry Quiz II. Matching _______1) Red in litmus _______2) [OH-] > [H+] _______3) pOH = 6.3 _______4) NaNO3(aq) _______5) KC2H3O2 (aq) _______6) pH = 5.2 _______7) pink in phenolphthalein _______8) vinegar _______9) tastes sour _______10) [OH-] = [H+] _______11) [H+] = 3.4 X 10-9 _______12) NH4Cl(aq) a) acid b) base c) neutral III. For each of the following reactions label the acid (a), the base (b), the conjugate acid (ca), and the conjugate base (cb). OH_______ 1) H2CO3 _______ + 2) CH3NH2 ______ + H2O _____ 3) HBr + ______ H2O ______ HCO3 _______ CH3NH3+ + _____ H3O + ______ + H2O _______ OH_____ + Br______ IV. Problems. You must show your work for credit. 1) Given [OH-] = 1.5 x 10 –5, find [H+] , pH , pOH and tell if it is acidic or basic. [H +] = pH = pOH = acidic or basic 2) Given pH = 5.7, find [OH-] , pOH , [H+] and tell if it is acidic or basic [OH -] = pOH = [H+] = acidic or basic 3) Find the pH of a 0. 0045 M solution of NaOH. 3) 4) The pH of a 0.063 M solution of hypobromous acid (HBrO) is 4.95. Calculate Ka. 4) 5) How many mL of a 0.34 M of HCl are needed to neutralize 125 mL of a 0.11 M Ba(OH)2 ? 5) 6) What is the molar concentration of15 ml H3PO4 if it takes 25 mL of 0.85 M NaOH to neutralize the acid ? 6) 7 How many equilivants are in 6.34 L of a 0.75N solution of H3PO4 7) 8a) ________ Mg(OH)2 + ________ HBr 8b) ________ Al(OH)3 + ______ H2CO3 9 If 25.00 mL of citric acid solution is titrated with 28.12 mL of 0.1718 N KOH, what is the concentration of citric acid? (hint: normality is a concentration unit!) 10)