Lab Handout
advertisement

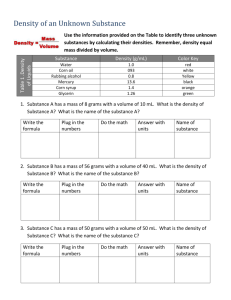
The Effect of Volume on the Density of a Substance EXPERIMENT (25 points) Observation: Density is a physical property of matter. Question: How does the volume of a substance (solid, liquid, gas) affect its density? Research / Review: VOLUME 1. How do you find the volume of a solid rectangular block? 2. How do you find the volume of an irregularly shaped object? 3. How do you find the volume of a liquid? 4. What are the two measurement units for volume? 5. Review the procedure for determining the volume of a gas – make notes here. MASS 6. What are the mass measurement units? 7. Review the procedure for finding the mass of a gas – make notes here. DENSITY 8. What is the equation for density? VARIABLE IDENTIFICATION 9. What is the dependent variable in this lab? 10. What is the independent variable in this lab? Collaboration: Decide which role you will play during this experiment. 1. Lab Manager: coordinates group members, gets and returns lab equipment, makes sure the lab table is clean when the experiment is completed, directs the analysis of data and formation of a conclusion that will answer the lab question. 2. Lab Technician(s): works with the group to write detailed lab procedures, performs the experiment, makes sure every group member understands how to measure mass and volume for each type of substance. 3. Data Analyst: works with the group to design an organized data table, records data during the experiment, calculates density, makes sure every group member understands how to calculate density. Hypothesis: Propose a hypothesis that predicts what will happen in your experiment. It should try to answer the lab question and reference what you will SEE happen during your experiment. If [how the independent variable will be changed], then [expected change in the dependent variable]. Experiment: Materials: Design your experiment using these materials. Apparatus: balance, 10 ml graduated cylinder, 50 ml graduated cylinder, gas collection apparatus, ruler Substances: Various regular solids, irregular solids, liquids and gases are provided. Each is labeled with a letter. Decide what substances you will use during your experiment to completely answer the lab question. Procedure: Write a detailed lab procedure so that your experiment could be easily replicated. Data: Decide what data you will collect. Create organized data tables for solid, liquid and gas. Be sure to include measurement units. When you are finished, create a graph to visually represent the data. Conclusion: In one paragraph address the following: Introduce the concept and the purpose and design of the experiment. (Lab Technician) Present the data and draw a clear conclusion that completely answers the lab question. (Data Analyst) State and examine the hypothesis – did the data support it? Suggest possible sources of error if it was not supported as well as next steps if needed. (Lab Manager)