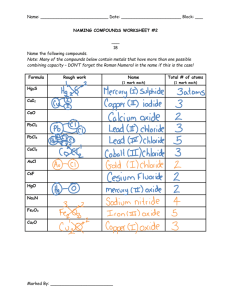
Writing Formulas Usi..
advertisement

The crossover method is a shortcut way of writing formulas for ionic compounds if you know the charges on the ions in the compound. We’ll go over a few examples a) Write the formula for magnesium chloride 2+ Mg – Cl MgCl2 In example (a), we’re asked to write the formula for magnesium chloride. a) Write the formula for magnesium chloride 2+ Mg – Cl MgCl2 Looking up Magnesium on the ion table or periodic table, we see its Mg with charge of 2+. a) Write the formula for magnesium chloride 2+ Mg – Cl MgCl2 And chloride is Cl with a charge of minus, or minus 1. a) Write the formula for magnesium chloride 2+ Mg – Cl Mg1Cl2 We start the formula by writing down the symbols, Mg and Cl a) Write the formula for magnesium chloride 2+ Mg – Cl Mg1Cl2 Now we take the 2 from the charge on magnesium, (click) cross over, and (click) write it to next to the chloride. a) Write the formula for magnesium chloride 2+ Mg – Cl Mg1Cl2 Now we could take the 1 from the charge on chloride, (click) cross over, and write it next to the magnesium, but we don’t use the subscript 1 in a formula, so we don’t need to write anything. a) Write the formula for magnesium chloride 2+ Mg Mg1Cl2 So the formula has one Mg and two Cl’s. – Cl a) Write the formula for magnesium chloride 2+ Mg – Cl MgCl2 Formula So we’ll compact it and write the final formula for magnesium chloride as MgCl2. a) Write the formula for magnesium chloride 2+ Mg – 2Cl MgCl2 Formula We’ll check the charges to verify that this formula is correct. (click) We have one Mg2+ ion and 2 Cl– ions a) Write the formula for magnesium chloride +2 2+ Mg + 2(–1) – 2Cl MgCl2 Formula So the total charge is positive 2, plus 2 × –1, or positive 2 plus negative 2, a) Write the formula for magnesium chloride +2 2+ Mg + 2(–1) – 2Cl MgCl2 Formula Which is equal to zero, so the formula MgCl2 is correct. = 0 b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 Example b asks us to write the formula for calcium oxide. b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 We look up calcium and its Ca with a charge of positive 2. b) Write the formula for calcium oxide 2+ Ca Ca2O2 And oxide is “O” with a charge of negative 2. 2– O b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 We start the formula by writing down the symbols Ca and O. b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 The charge on calcium is positive 2, so we (click) cross over and (click) write a 2 next to Oxygen in the formula b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 The charge on oxide is negative 2, so we (click) cross over and (click) write a 2 next to Calcium in the formula. Notice we always drop the negative when we write subscripts b) Write the formula for calcium oxide 2+ Ca Ca2O2 Now, we have the formula Ca2O2. 2– O b) Write the formula for calcium oxide 2+ Ca 2– O Ca2O2 Both subscripts can be divided by 2 But you can see that both of the subscripts on this formula are divisible by 2. b) Write the formula for calcium oxide 2+ Ca 2– O Ca12O21 Divide both subscripts by 2 So we’ll simplify and divide both of these by 2 and get 1’s b) Write the formula for calcium oxide 2+ Ca 2– O Ca12O21 Remove the 1’s We do not keep the subscript 1 in a formula, so we (click) remove the 1’s, leaving us with the formula CaO. b) Write the formula for calcium oxide 2+ Ca 2– O CaO2 Formula Which we’ll compact and write as the final formula for calcium oxide. b) Write the formula for calcium oxide 2+ Ca 2– O CaO2 Formula Checking charges, we have one Ca2+ ion and one O2– ion. b) Write the formula for calcium oxide +2 2+ Ca + CaO2 Formula So the total charge is positive 2 plus negative 2, –2 2– O b) Write the formula for calcium oxide +2 2+ Ca + CaO2 Formula Which is equal to zero. So this formula is correct. –2 = 0 2– O c) Write the formula for chromium(III) sulphide 3+ Cr 2– S Cr2S32 Question c asks us to write the formula for chromium(III) sulphide. c) Write the formula for chromium(III) sulphide 3+ Cr Cr2S32 The chromium (III) ion is Cr 3 plus 2– S c) Write the formula for chromium(III) sulphide 3+ Cr Cr2S32 And the sulphide ion is S 2 minus 2– S c) Write the formula for chromium(III) sulphide 3+ Cr Cr2S32 We start the formula with the symbols Cr and S 2– S c) Write the formula for chromium(III) sulphide 3+ Cr 2– S Cr2S32 We take the 3 from the charge on chromium, (click) cross over and write (click) 3 next to sulphur in the formula. c) Write the formula for chromium(III) sulphide 3+ Cr 2– S Cr2S32 And we take the 2 from the 2 minus charge on the sulphide ion and (click) cross over and write (click) 2 next to chromium in the formula. c) Write the formula for chromium(III) sulphide 3+ Cr 2– S Cr2S32 There is no number other than 1, that will divide into 2 and 3, so this formula cannot be simplified. c) Write the formula for chromium(III) sulphide 3+ Cr 2– S Cr2S32 Formula So the final formula for chromium III sulphide is Cr2S3. c) Write the formula for chromium(III) sulphide 3+ 2Cr 2– 3S Cr2S32 Formula Checking charges, we have 2 Cr3+ ions and 3 S2– ions, c) Write the formula for chromium(III) sulphide 2(+3) 3+ 2Cr + 3(–2) 2– 3S Cr2S32 Formula So the total charge is 2 × positive 3, plus 3 × (–2), or positive 6 plus –6, c) Write the formula for chromium(III) sulphide 2(+3) 3+ 2Cr + 3(–2) 2– 3S Cr2S32 Formula Which is equal to zero, so this formula is correct. = 0 d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 Question d asks us to write the formula for palladium(IV) oxide. d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 Palladium(IV) is Pd with a charge of 4 plus d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 And oxide is “O” with a 2 minus charge d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 We start the formula with the symbols Pd and O d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 We take the 4 from the charge on palladium, (click) crossover and (click) write 4 next to oxygen in the formula d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 We take the 2 from the charge on the oxide ion, (click) crossover and (click) write 2 next to palladium in the formula d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 Now, we have the formula Pd2O4. d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd2O42 Both subscripts can be divided by 2 But we see that both of the subscripts 2 and 4 are divisible by 2. d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd12O42 2 Divide both subscripts by 2 So we simplify by dividing both the 2 and the 4 by 2, giving us 1 and 2. d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd12O42 2 Remove the 1 We remove the subscript 1 d) Write the formula for palladium(IV) oxide 4+ Pd 2– O Pd12O42 2 Leaving us with the formula PdO2. d) Write the formula for palladium(IV) oxide 4+ Pd 2– O PdO2 Formula Which we’ll compact and state as the final formula for palladium(IV) oxide d) Write the formula for palladium(IV) oxide 4+ Pd 2– 2O PdO2 Formula Checking charges, we have one Pd4+ ion and two O2– ions d) Write the formula for palladium(IV) oxide +4 4+ Pd + 2(–2) 2– 2O PdO2 Formula So the total charge is positive 4 plus 2 times –2, or positive 4 plus negative 4, d) Write the formula for palladium(IV) oxide +4 4+ Pd + 2(–2) = 0 2– 2O PdO2 Formula Which is equal to zero, so PdO2 is the correct formula.