Aldehydes and Ketones: Organic Chemistry Presentation
advertisement

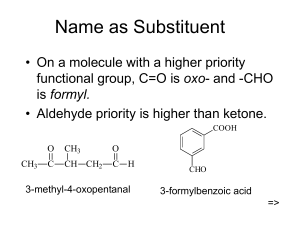
ORGANIC CHEMISTRY || T.A HAYA S ALSUBAIE Pharmaceutical chemistry department Pharmacy college Princess Nora University ALDEHYDES AND KETONES OBJECTIVES Write the functional groups of aldehydes and ketones. Determine chemical and physical properties of aldehydes and ketones. Perform chemical tests to distinguish between aldehydes and ketones. INTRODUCTION Aldehydes and ketones are simple compounds which contain a carbonyl group . (c=o) (R=alkyl or aryl ) Aldehyde • Has at least one hydrogen atom attached directly to carbonyl carbon. • Hydrogen atom makes aldehydes very easy to oxidize . ketone • Has two alkyl(or aryl) groups attached to carbonyl carbon. • Ketones don't have that particular hydrogen atom, resistant to oxidation. • Only very strong oxidizing agents like (potassium permanganate solution) oxidize ketones. PHYSICAL PROPERTIES OF ALDEHYDE Formaldehyde & Acetaldehyde State Liquid color colorless odor pungent odor flammability Blue non-smoky flame solubility completely miscible with water effect on LP neutral structure formaldehyde acetaldehyde PHYSICAL PROPERTIES OF KETONES Acetone State Liquid color colorless Odor characteristic odor Flammability Blue non-smoky flame Soloubility completely miscible with water effect on L.P Neutral Structure Chemical test Specific test General test Oxidation test Tollen’s test Schiff’s reagent Nanitroprussid Fehling test Iodoform test Salicylic acid test CHEMICAL TESTS General Test: OXIDATION TESTS: 1-Tollens’ silver mirror test: procedure: 1 ml of ald. or ket. + 1 ml of Tollens’ reagent ;then warm in water bath for 2-5 min. ALDEHYDE SILVER Mirror KETONE NO Reaction TOLLENS’ SILVER MIRROR TEST: Acetaldehyde Acetone Formaldehyde CHEMICAL TESTS General Test: OXIDATION TESTS: 2) Fehling test: procedure: 1 ml of ald. or ket. + 1 ml of Fehling reagent A&B then warm in water bath for 2-5 min. aldehyde Rapidly change in blue color Acetone No reaction FEHLING TEST Acetaldehyde Formaldehyde Acetone CHEMICAL TESTS General Test: Schiff test: procedure: 1 ml of ald. or ket. + 2 drops of Schiff reagent. Aldehyde Ketone Magenta color Color slightly appear SCHIFF TEST: Acetone Acetaldehyde Formaldehyde CHEMICAL TESTS Specific tests: 1) Na-nitroprussid: procedure: 1 ml of ald. or ket. + 1 ml of Na- nitroprussid + 1 ml of 10% NaOH. Formaldehyde Acetaldehyde Acetone No reaction Red color Red color NA-NITROPRUSSID: Acetone Acetaldehyde Formaldehyde CHEMICAL TESTS: Specific tests: Iodoform test: procedure: 1 ml of ald. or ket. + 3 ml of 30% NaOH+ 1 ml of I2 solution then warm in water bath for 2-5 min.( add more than 1 ml of I2 if you need ) Formaldehyde No reaction Acetaldehyde Yellow ppt. acetone Yellow ppt. IODOFORM TEST Acetaldehyde Formaldehyde Acetone CHEMICAL TESTS: Specific tests: Salicylic acid : procedure: 1 ml of formaldehyde + few amount of salicylic acid + 2 drops of conc. H2SO4. Formaldehyde Crimson red color SALICYLIC ACID TEST Formaldehyde PRE-LAB QUESTIONS Give a structure of a compound that contains the following functional groups: - Aldehyde - Ketone Why can distinguish between an aldehyde and a ketone using Tollen’s Reagent ? LAB SAFETY THANK YOU ..