Chemistry Semester Test Study Guide Chapters 5
advertisement

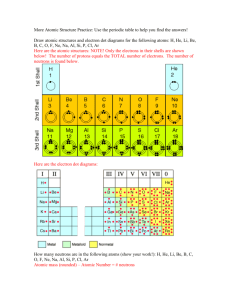
Chemistry Semester Test Study Guide Chapters 5-7 electronegativity ionization energy atomic radius metal transition metal periodic law cation group period electron halide ion octet rule ionic bond electron dot structure valence electron coordination number metallic bond principal quantum number orbital sublevel In Bohr’s model of the atom, where are the electrons and protons located? How many sublevels are in each principal energy level? How many orbitals are in the s, p, d, and f sublevels? How many electrons can be in an orbital? In terms of energy, what happens to an electron when it jumps to a higher energy level? How do electrons spin in an orbital? What does the Heisenberg Uncertainty Principle state? Who arranged the elements according to atomic mass? How is the modern day periodic table arranged? Where are the metals, nonmetals, and metalloids located on the periodic table? How does atomic radius differ from the top to bottom of a group on the periodic table? What is the element with the highest electronegativity value? How many valence electrons are in an atom of phosphorus? What is the electron configuration for the calcium ion? How many electrons does barium have to give up to achieve noble gas electron configuration? What is the charge on the cation in sodium sulfide? What is the formula unit of sodium nitride? What is the formula for sodium sulfate? Ionic compounds are normally in which physical state at room temperature? Under what conditions can potassium bromide conduct electricity? What characteristic of metals make them good conductors of electricity? How many electrons are there in the highest occupied energy level of a phosphorus atom? What is the formula formed when potassium reaches noble gas electron configuration?