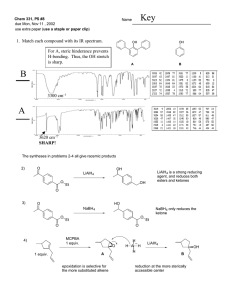
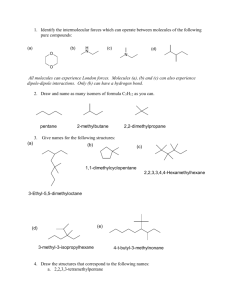
Revision
advertisement

Winning Fifty Points Revision 430 Chem. What is the IUPAC name for (CH3)3CCH(OH)CH2CH(OH)CH2CH(CH3)2? A) 1,1,1,6-tetramethyl-2,4-heptanediol B) 2,2,7-trimethyl-3,5-octanediol C) 2,2,6,6-tetramethyl-3,5-heptanediol D) 2,7,7-trimethyl-4,6-octanediol Which of the following reagents would you expect to react with bromocyclopentane by an SN2 mechanism? A) C2H5OH B) C2H5O(-) K(+) C) NaCN D) (CH3)3N Compound X (C6H12) reacts with HI. The product of this reaction, when treated with KOH in ethanol, gives Y ( an isomer of X ). Ozonolysis of X (H2O2 workup) produces two compounds: a two carbon Aldehyde, and a four carbon ketone. What is X? A) 2-methyl-2-pentene B) 4-methyl-1-pentene C) 2,3-dimethyl-2-butene D) 3-methyl-1-pentene Which one of the following alcohols will be oxidized by (CrO3 in 50% sulphuric acid) to a ketone having the same number of carbon atoms ? A) 1-methylcyclohexanol B) 3,3-dimethylcyclopentanol C) 3-methyl-1-hexanol D) 3-ethyl-3-hexanol Which reagent(s) would best accomplish the following transformation A) H3O+ & heat B) (i) HgSO4 in H2O (ii) NaBH4 C) (i) B2H6 in ether (ii) H2O2 and base D) (i) HOBr (ii) Mg in ether Which of the following reagents would not effect the following transformation? A) HCl & ZnCl2 B) PCl3 C) SOCl2 D) KCl (5 molar solution) A C7H13Br compound reacts with KOH in ethanol to form 3methylcyclohexene as the major product. What is a likely structure for the starting alkyl bromide? A) cis-4-methylcyclohexyl bromide B) trans-3-methylcyclohexyl bromide C) cis-2-methylcyclohexyl bromide D) trans-2-methylcyclohexyl bromide Synthesis of hexane-3,4-diol from trans-3-hexene may be accomplished in two ways: (i) OsO4 hydroxylation & (ii) C6H5CO3H epoxidation followed by NaOH opening of the epoxide ring. Which of the following statements about the products from these reactions is correct? A) the two methods give the same product B) (i) gives a chiral isomer, (ii) gives an achiral isomer C) (i) gives an achiral isomer, (ii) gives a chiral isomer D) two different isomers are formed, but both are chiral Which of the following reaction sequences would best serve to convert 2-methyl-1bromopropane to 4-methyl-1-iodopentane? A) (i) Mg in ether; .(ii) ethylene oxide (C2H4O); (iii) HI & heat B) (i) NaC≡CH in ether; .(ii) H2 + Lindlar catalyst; (iii) HI C) (i) KOH in alcohol; . (ii) C6H5CO3H in CH2Cl2; (iii) NaC≡CH in ether ; (iv) 2 H2 + Pt catalyst D) (i) NaC≡CH in ether; .(ii) H3O+ + HgSO4; (iii) HI & heat Which of the following reagents would be best for oxidizing a 1º-alcohol to an aldehyde? A) H3PO4 B) POCl3 in CH2Cl2 C) Jones' reagent (H2CrO4))or orKMnO KMnO44// HH++ D) OsO4