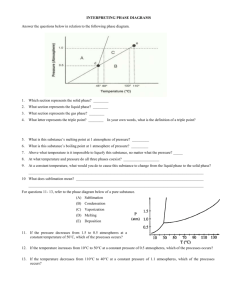
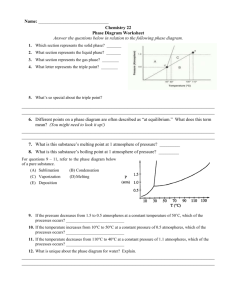
Substance absorbs heat energy Melting
advertisement

Topic: Chemistry Aim: Explain the different types of phase changes and the energy changes involved. Do Now: ON NOTES SHEET HW: Ditto – Periodic Tables Families Review (last page of Periodic Table Notes Packet) http://www.youtube.com/watch?v=XaDxwe8l3us&feature=related Phase Changes • Involves the absorption OR release of heat (energy). Substance absorbs heat energy Melting • Solid liquid • Solid ABSORBS heat energy Substance absorbs heat energy Melting points of different substances • Iron: 1536ºC • Gold: 1063ºC • Glucose: 150ºC • Urea: 132-134ºC Vaporization • • • Liquid gas Liquid ABSORBS heat energy When it occurs on surface of liquid: evaporation GAS Liquid absorbs heat energy LIQUID Boiling points of different substances • Alcohol (Ethanol): 79ºC • Iodine: 184.3ºC • Mercury: 356.9ºC • Olive oil: 300ºC • Jet fuel: 163ºC Condensation • Gas liquid • Gas LOSES heat energy GAS Gas loses heat energy LIQUID Freezing • Liquid solid • Liquid LOSES heat energy Substance loses heat energy Sublimation • Solid Gas • No liquid phase • Ex: dry ice GAS Solid absorbs heat energy Solid Moth balls commonly undergo the process of sublimation. Sublimation is the process in which a solid changes directly to its gaseous phase, skipping the liquid phase. And an animation of the phase changes of water Let’s summarize… 1. List the different types of phase changes. 2. Explain what happens to heat energy in each phase change. 1. When a liquid becomes a solid, energy 1. does not change 2. is released 3. is absorbed 4. is first absorbed, then released 2. _______ is the process in which liquid changes to gas below the boiling point. 1. Sublimation 2. Condensation 3. Evaporation 4. Combustion 3. On a hot day, the outside of a glass containing a cold drink becomes frosty. When touching the glass, it feels 1. water that has passed through the glass 2. water that has condensed from a gas in the air 3. water that has solidified 4. water that has absorbed energy 4. The change of a liquid to a solid is called 1. freezing 2. Melting 3. sublimation 4. vaporization 5. During which of the following processes is there a decrease in the heat content of the form of water indicated? 1. Ice as it forms on a lake 2. Water droplets as they fall to the ground 3. Water as it evaporates from a pond 4. Snow as it melts on a mountainside 6. When substances go directly from the solid phase to the gas phase, the phase change is called 1. sublimation 2. condensation 3. evaporation 4. vaporization E D condensing 100 Temperature º C. boiling C B freezing 0 melting A Heat (thermal energy) • http://www.youtube.com/watch?v=vDZhUk p30tE&feature=related