sathyabama university

Register Number
SATHYABAMA UNIVERSITY
(Established under section 3 of UGC Act,1956)
Course & Branch :B.E/B.Tech - P-CHEM/P-MECH
Title of the Paper :Material Technology Max. Marks:80
Sub. Code :SMEX1006(2010-11-12) Time : 3 Hours
Date :31/05/2012 Session :FN
______________________________________________________________________________________________________________________
PART - A (10 x 2 = 20)
Answer ALL the Questions
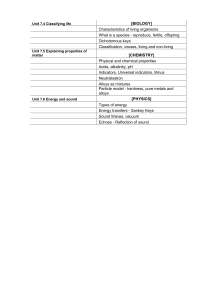
1. What is Gibbs phase rule?
2. Define Eutectoid reaction.
3. Give general composition of brazing alloy.
4. What is meant by grey cast iron?
5. What are the various types of surface hardening process?
6. Why are annealing processes carried out?
7. Write any four characteristics of metal powders.
8. What do you mean by powder metallurgy?
9. Draw a typical S N curve for Mild steel.
10. Mention two factors affecting fatigue.
PART – B (5 x 12 = 60)
Answer All the Questions
11. Find the Milller Indices and show graphically for the following intercepts:
(2,4,3), (2,4,8), (0.5,1.5,2.5), (4.5,7.5,9), (1,1/0,1/0).
(or)
12. Draw the equilibrium for two metals completely miscible in the liquid state and partially miscible in the solid state. Show how the diagram can be use to predict the equilibrium proportions of phases present for two alloys.sketch the microstructures you would obtain in the two alloys chosen.
13. Discuss the applications of Aluminium and Aluminium alloys.
(or)
14. Discuss the major composition of Cupper & its alloys.
15. What do you understand by the term hardenability and explain how it is determined? Explain the process involved in sequence.
(or)
16. Compare the different methods of Surface hardening with respect to application, advantages and limitations.
17. Contrast mechanical and hydraulic compacting presses with regard to advantages, disadvantages and applications.
(or)
18. Explain electro chemical method of production of metallic powder and discuss the process parameters of the method.
19. Explain (a) Modes of Fracture (b) Cup and cone fracture.
(or)
20. Explain the various stages of creep failure with a neat sketch.