PPT: GAS LAWS
advertisement

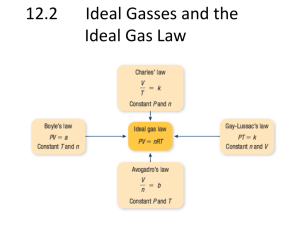
Gas Laws Characteristics of Gases • highly compressible. • occupy the full volume of their containers. • exert a uniform pressure on all inner surfaces of a container • diffuse (mix) easily and quickly • have very low densities. Kinetic Molecular Theory – Gases consist of a large number of molecules in constant random motion. – Volume of individual molecules negligible compared to volume of container. – Intermolecular forces (forces between gas molecules) negligible. – Collision of gas particles are elastic so no kinetic energy is lost – As temperature increases the gas particles move faster, hence increased kinetic energy. Four Physical Quantities for Gases Phys. Qty. Symbol SI unit Other common units pressure P Pascal (Pa) atm, mm Hg, torr, psi volume V m3 dm3, L, mL, cm3 temp. T K °C, °F moles n mol Temperature Always use absolute temperature (Kelvin) when working with gases. ºF -459 ºC -273 K 0 C F 32 5 9 32 212 0 100 273 373 K = ºC + 273 Practise Absolute zero is – 273C or 0 K Calculate the missing temperatures 0C = _______ K 100C = _______ K 100 K = _______ C – 30C = _______ K 300 K = _______ C 403 K = _______ C 25C = _______ K 0K = _______ C Kelvin Practice What is the approximate temperature for absolute zero in degrees Celsius and kelvin? Absolute zero is – 273C or 0 K Calculate the missing temperatures 273 K 373 K 0C = _______ 100C = _______ – 173 C 100 K = _______ 243 K – 30C = _______ 300 K = _______ C 27 403 K = _______ 130 C 298 K 25C = _______ 0K – 273 C = _______ Pressure Pressure (P ) is defined as the force exerted per unit area. The atmospheric pressure is measured using a barometer. force pressure area Which shoes create the most pressure? Pressure KEY UNITS AT SEA LEVEL 101.325 kPa (kilopascal) 1 atm 760 mm Hg 760 torr N kPa 2 m 14.7 psi •1 atm = 760 mmHg = 760 torr = 101325 Pa. Pressure Barometer • measures atmospheric pressure Aneroid Barometer Mercury Barometer STP STP Standard Temperature & Pressure 273 K 101.325 kPa STP SLC Standard Laboratory Conditions 25°C or 298 K 101.325 kPa The Gas Laws -BOYLES -CHARLE -GAY-LUSSAC Boyle’s Law The pressure and volume of a gas are inversely related at constant mass & temp. PV = k P V P1 V1 P2 V2 A. Boyle’s Law Practise A sample of chlorine gas occupies a volume of 946 mL at a pressure of 726 mmHg. What is the pressure of the gas (in mmHg) if the volume is reduced at constant temperature to 154 mL? A sample of chlorine gas occupies a volume of 946 mL at a pressure of 726 mmHg. What is the pressure of the gas (in mmHg) if the volume is reduced at constant temperature to 154 mL? P1 x V1 = P2 x V2 P2 = P1 = 726 mmHg P2 = ? V1 = 946 mL V2 = 154 mL P1 x V1 V2 726 mmHg x 946 mL = = 4460 mmHg 154 mL Charles’ Law The volume and absolute temperature (K) of a gas are directly related at constant mass & pressure V k T V T V1 V2 T1 T2 Charles’ Law Practise 2. A sample of gas occupies 3.5 L at 300 K. What volume will it occupy at 200 K? 3. If a 1 L balloon is heated from 22°C to 100°C, what will its new volume be? 2. A sample of gas occupies 3.5 L at 300 K. What volume will it occupy at 200 K? V1 = 3.5 L, T1 = 300K, V2 = ?, T2 = 200K 3.5 L / 300 K = V2 / 200 K V2 = (3.5 L/300 K) x (200 K) = 2.3 L 3. If a 1 L balloon is heated from 22°C to 100°C, what will its new volume be? V1 = 1 L, T1 = 22°C = 295 K V2 = ?, T2 = 100 °C = 373 K V1/T1 = V2/T2, 1 L / 295 K = V2 / 373 K V2 = (1 L/295 K) x (373 K) = 1.26 L For more lessons, visit www.chalkbored.com A sample of carbon monoxide gas occupies 3.20 L at 125 0C. At what temperature will the gas occupy a volume of 1.54 L if the pressure remains constant? V1/T1 = V2/T2 T2 = V1 = 3.20 L V2 = 1.54 L T1 = 398.15 K T2 = ? V2 x T1 V1 = 1.54 L x 398.15 K 3.20 L = 192 K Gay-Lussac’s Law The pressure and absolute temperature (K) of a gas are directly related at constant mass & volume P k T P T Gay-Lussac’s Law Combined Gas Law P 1V 1 T1 = P 2V 2 T2 P 1 V 1T 2 = P 2V 2 T 1 E. Gas Law Problems gas occupies 473 cm3 at 36°C. Find its volume at 94°C. A CHARLES’ LAW GIVEN: T V V1 = 473 cm3 T1 = 36°C = 309K V2 = ? T2 = 94°C = 367K WORK: P1V1T2 = P2V2T1 (473 cm3)(367 K)=V2(309 K) V2 = 562 cm3 E. Gas Law Problems A gas occupies 100. mL at 150. kPa. Find its volume at 200. kPa. BOYLE’S LAW GIVEN: P V V1 = 100. mL P1 = 150. kPa V2 = ? P2 = 200. kPa WORK: P1V1T2 = P2V2T1 (150.kPa)(100.mL)=(200.kPa)V2 V2 = 75.0 mL Practise occupies 7.84 cm3 at 71.8 kPa & 25°C. Find its volume at STP. A gas E. Gas Law Problems gas occupies 7.84 cm3 at 71.8 kPa & 25°C. Find its volume at STP. A COMBINED GAS LAW GIVEN: P T V WORK: V1 = 7.84 cm3 P1V1T2 = P2V2T1 P1 = 71.8 kPa (71.8 kPa)(7.84 cm3)(273 K) T1 = 25°C = 298 K =(101.325 kPa) V2 (298 K) V2 = ? P2 = 101.325 kPa V2 = 5.09 cm3 T2 = 273 K E. Gas Law Problems A gas’ pressure is 765 torr at 23°C. At what temperature will the pressure be 560. torr? GAY-LUSSAC’S LAW GIVEN: P T WORK: P1 = 765 torr P1V1T2 = P2V2T1 T1 = 23°C = 296K (765 torr)T2 = (560. torr)(309K) P2 = 560. torr T2 = 226 K = -47°C T2 = ? Avogadro’s Principle Equal volumes of all gases contain equal numbers of moles at constant temp & pressure. V k n V n V1 V2 n1 n2 11.5 The Ideal Gas Equation The gas laws can be combined into a general equation that describes the physical behavior of all gases. 1 V P V T V n Boyle’s law Charles’s law Avogadro’s law nT V P nT V R P rearrangement PV = nRT R is the proportionality constant, called the gas constant. B. Ideal Gas Law PV=nRT UNIVERSAL GAS CONSTANT R = 8.3145 J/mol·K R=0.0821 Latm/molK R = 0.0821 liter·atm/mol·K R = 8.3145 J/mol·K R = 8.2057 m3·atm/mol·K R = 62.3637 L·Torr/mol·K or L·mmHg/mol·K B. Ideal Gas Law Calculate the pressure in atmospheres of 0.412 mol of He at 16°C & occupying 3.25 L. IDEAL GAS LAW GIVEN: WORK: P = ? atm PV = nRT n = 0.412 mol P(3.25)=(0.412)(0.0821)(289) L mol Latm/molK K T = 16°C = 289 K V = 3.25 L P = 3.01 atm R = 0.0821Latm/molK B. Ideal Gas Law Find the volume of 85 g of O2 at 25°C and 104.5 kPa. IDEAL GAS LAW GIVEN: WORK: V=? 85 g 1 mol = 2.7 mol n = 85 g = 2.7 mol 32.00 g T = 25°C = 298 K PV = nRT P = 104.5 kPa (104.5)V=(2.7) (8.315) (298) kPa mol dm3kPa/molK K R = 8.315 dm3kPa/molK V = 64 dm3 Summary Dalton found that the total pressure of mixed gases is equal to the sum of their individual pressures (provided the gases do not react). 50 kPa 100 kPa 150 kPa Note: all of these + = volumes are the 1 L oxygen 1 L nitrogen 1 L mixed gas same Gas Mixtures Each component of a gas mixture exerts a pressure independent of the other components. The total pressure is the sum of the partial pressures. Practise • Calculating partial pressures Vapour Pressure Defined • Vapour pressure is the pressure exerted by a vapour. E.g. the H2O(g) in a sealed container. Eventually the air above the water is filled with vapour pushing down. As temperature , more molecules fill the air, and vapour pressure . Collecting gases over water • Many times gases are collected over H2O • Often we want to know the volume of dry gas at STP (useful for stoichiometry). For this we must make 3 corrections: 1. The level of water inside and outside the tube must be level (so pressure inside is equal to the pressure outside). 2. The water vapour pressure must be subtracted from the total pressure (to get the pressure of the dry gas). 3. Finally, values are converted to STP using the combined gas law. Sample calculation A gas was collected over 21°C H2O. After equalizing water levels, the volume was 325 mL. Give the volume of dry gas at STP (Patm=102.9 kPa). Step 1: Determine vapour pressure (pg. 464) At 21°C vapour pressure is 2.49 kPa Step 2: Calculate the pressure of dry gas Pgas = Patm - PH2O = 102.9 - 2.49 = 100.41 kPa Step 3: List all of the data T1 = 294 K, V1 = 325 mL, P1 = 100.41 kPa Step 4: Convert to STP = 299 mL (P1)(V1)(T2) (100.4 kPa)(325 mL)(273 K) V2= = (P2)(T1) (101.325 kPa)(294 K) Assignment 1. 37.8 mL of O2 is collected by the downward displacement of water at 24°C and an atmospheric pressure of 102.4 kPa. What is the volume of dry oxygen measured at STP? 2. Try questions 8 – 10 on page 465. 3. 236 mL of H2 is collected over water at 22°C and at an atmospheric pressure of 99.8 kPa. What is the volume of dry H2 at STP? 4. If H2 is collected over water at 22°C and an atmospheric pressure of 100.8 kPa, what is the partial pressure of the H2 when the water level inside the gas bottle is equal to the water level outside the bottle? 1) Vapor pressure at 24C = 2.98 kPa Pgas = Patm - Pvapor = 102.4 kPa - 2.98 kPa = 99.42 kPa = P1 V1 = 37.8 mL, P1 = 99.42 kPa, T1 = 297 K V2 = ?, P2 = 101.3 kPa, T2 = 273 K P1V1 = T1 (99.42 kPa)(37.8 mL) = (297 K) P2V2 T2 (101.3 kPa)(V2) (273 K) (99.42 kPa)(37.8 mL)(273 K) (V2) = = 34.1 mL (297 K)(101.3 kPa) 3) Vapor pressure at 22C = 2.64 kPa Pgas = Patm - Pvapor = 99.8 kPa - 2.64 kPa = 97.16 kPa = P1 V1 = 236 mL, P1 = 97.16 kPa, T1 = 295 K V2 = ?, P2 = 101.3 kPa, T2 = 273 K P1V1 P2V2 = T1 T2 (97.16 kPa)(236 mL) (101.3 kPa)(V2) = (295 K) (273 K) (97.16 kPa)(236 mL)(273 K) (V2) = = 209 mL (295 K)(101.3 kPa) Answers 4 - Total pressure = PH2 + PH2O 100.8 kPa = PH2 + 2.64 kPa 100.8 kPa - 2.64 kPa = PH2 = 98.16 kPa For more lessons, visit www.chalkbored.com Ideal and real gas • Ideal gas: • Ideal gas molecule have - zero volume -zero attraction between molecules • Real gases behave ideally at - high temperature - low pressure