unit 1 introduction to basic biochemistry
advertisement

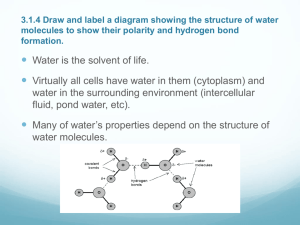
PRT3402- Agricultural Biochemistry PJJ UPM / UPMET UNIT 1 INTRODUCTION TO BASIC BIOCHEMISTRY Introduction to Unit The main purpose of this course is to give an understanding of the basic principles and concepts of biochemistry in relation to agriculture thus the name of the course – Agricultural Biochemistry. The main thrust of each topic will be on biochemistry but will draw examples mainly from the field of agriculture. Since biochemistry is the study of the biological processes of life at the molecular level, this initial unit will expose students to some key components or elements involve in biochemistry. It starts with the description of cellular and organizational structure of eukaryotic and prokaryotic cells, animal and plant cell, followed by water properties (the main media for biochemical processes), types of chemical reactions, acidity, alkalinity and buffers in aqueous solutions. Learning Outcomes At the end of this unit the students will be able to: 1. Have a general overview of the course 2. Describe the topics that will be discuss in this course 3. Differentiate animal and plant cell structures 4. Explain water properties and its relationship to chemical reactions involving acids, bases and buffers 1 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET Main Points 1. Unit 1 is a basic introduction to biochemistry starting with the cellular organization from eukaryote to prokaryote and differences between animal cells, an understanding of water properties, different chemical bonding and the basic explanation on acidity, alkalinity and buffers in aqueous solutions. The students will learn the differences between eukaryote and prokaryotes, animal and plant cell structure and appreciate the importance of water as the main media for all biochemical reactions. In addition the pH of an aqueous solution and how it affects biochemical reactions will also be studied. 2. In Unit 2, you will learn about the structure and function of amino acids and how amino acids are arranged to form polypeptides and then made into proteins; different structures of the proteins will be described (primary, secondary, tertiary and quarternary structures) and some examples of proteins functions are explained using collagen, hemoglobin and immunoglobin as examples. 3. Unit 3 will give you a description of the role of carbohydrates starting with simple sugars which form monosaccharides. From monosaccharides, several sugars are put together to form polysaccharides thus forming the group of oligosaccharides and polymers known as carbohydrates. 4. Unit 4 will describe the structure and function of lipids which is formed from fatty acids. The complex polymers of fatty acids which form triglycerides, phospholipids, sphingolipids, wax, terpenes and steroids will also be described. The function and structure of membranes will also be discussed in this Unit 4. It will include description of lipid and protein bilayer, how compounds and molecules are transported across membranes using active and passive transport mechanisms. 5. In Unit 5, you will be taught about the structure and function of nucleic acids, DNA and RNA which are the basic building blocks for genomic functions of a cell. Genomic function and organization and a description of 2 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET information flow from genome to cellular functions will be explained. You will also learn about the how genetic code is organized, how DNA replicates and a trait is inherited and how mutations arise in an organism which leads to different genotypes and phenotypes. 6. Topic of Unit 6 will focus on the function of enzymes which are biological catalysts. You will learn about enzyme characteristics of an enzyme, classification and nomenclature, kinetics, regulation of enzyme activities and how an enzyme assay is conducted. 7. Bioenergetics will be the focus of Unit 7 in which description of energy generation processes in the cells are discussed. This includes glycolysis and fermentation, citric acid or tricarboxylic (TCA) cycle, gluconeogenesis and the electron transport system. Mitochondria function and structure will also be discussed. 8. Energy and photosynthate generation by plants will be introduced in Unit 8. You will discuss photosynthesis, phosphorylation, Kelvin cycle and how photosynthesis is regulated under light dependent and light independent phases. 9. In the final Unit 9, discussion will help you to discover what are hormones, their effects on living cells and organisms, types of hormones, differences between exocrine and endocrine glands and the regulation of hormone reactions and mechanism. TOPIC 1 : CELL ORGANIZATION 3 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET Main Points 1.1 The fundamental unit of all living organisms is the cell. Cells are the component that make up the living organisms which can be unicellular (single cell) or multicellular. 1.2 Cells are very tiny (size ranging from 0.1 um to 200 um) and if observed under the microscope, the cellular structure can be classified into two basic types i.e. eukaryote and prokaryote cells. 1.3 Prokaryote have a size range of diameter from 0.1 um to 50 um (but the length can reach 0.5 mm) whereas eukaryote has a bigger size range of 2 to 200 um. Bacteria and Archea represents prokaryotic cell type where as fungi, algae and protozoa showed the eukaryotic cell type. Multicellular organisms (plants and animals) are also made-up of eukaryotic cells. 1.4 Structurally prokaryotes are simpler than eukaryotes. The most obvious difference between these cell types is the absence of nucleus and other cell organelles such as mitochondria and chloroplasts (plant and algal cells) in the prokaryotes.. 1.5 The prokaryotic cell can be presented by three main regions: (a) presence of flagella or/and pili on the outside; (b) cell is enclosed by a cell envelope and plasma membrane and (c) a cytoplasmic area containing the chromosomic DNA. The DNA in prokaryotes is usually arranged in a circular manner - nucleoid. Some bacteria have extrachromosomal DNA known as plasmids. 1.6 In eukaryotic cells, the outside can be ciliated or flagellated. The cilia functions as cellular sensor that coordinate signaling pathways. Eukaryotes can move using the cilia or flagella. 1.7 The plasma membrane is similar to prokaryotes. Cell wall may be present. 1.8 DNA which made-up the linear chromosomes surrounding histones are typical of eukaryotes. They are enclosed within the nucleus and membrane-bound within the cytoplasmic region. Mitochondria, Golgi 4 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET apparatus, lysosomes, ribosomes, vesicles, endoplasmic reticulum are some of the structures that may be present in the cytoplasm of eukaryotes. 1.9 A typical representation of prokaryote and eukaryote cell (plant and animal cell) is given below. Prokaryote Cell 1.1 Although plant and animal cells are both eukaryotic cells, there are several differences between the two. A plant cell typically have chloroplasts, large vacuoles and cell wall which is usually absent in animal cell. However, animal cell may contain microvilli, lysosomes and lipid droplets which are not present in plant cells. Eukaryote cells 5 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET Plant Cell 1.10 Animal Cell The common organelles in both animal and plant cells include nucleolus and nucleus, chromosomal material, smooth and rough endoplastic reticulum, golgi complex, mitochondria, peroxisome, vesicles and microtubules. 1.11 Cytoplasm is the interior part of eukaryotic cell surrounded by plasma membrane excluding the nucleus. It includeorganelles such as mitochondria and cytosol. 1.12 Cell/Plasma membrane is a semi-permeable barrier separating the external and internal environment of the cell. Mainly consists of proteins and complex polar lipids. The lipids exist as phospholipids in the form of a fluid bilayer interspersed with membrane proteins and glycoproteins. The membranes are heavily involved in transporting nutrients and inorganic ions via several mechanisms. 1.13 Nucleus contains chromosomes which are the package of genetic material containing heritable genetic information encoded by DNA (deoxyribonucleic acid). The DNA material is binded by histones made 6 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET of lysine- and arginine rich proteins giving structural stability and part of it are subpackaged into a very dense structure known as the nucleolus. 1.14 There are two types of endoplasmic reticulum (ER): 1) Rough ER coated or embedded with ribosome are sites for protein synthesis and membrane. 2) Vesicles carrying newly synthesized protein are located in the smooth ER (without ribosome). Vesicles transport protein from ER to Golgi apparatus. The aqueous portion of ER is known as lumen. 1.15 Golgi complex is a system consisting of membrane bound vesicles which serve as the cell sorting and processing site. -Located close to the nucleus and ER -Receive vesicles from ER -Process macromolecules ready for delivery to other cellular compartments. 1.16 Mitochondria are organelles surrounded by two membranes - the inner membrane is known as the matrix which contains many enzymes for energy metabolism. The mitochondria is the main factory and ultimate site for generating ATP (adenosine triphosphate) - the energy currency of all cells from biofuel molecules. 7 PRT3402- Agricultural Biochemistry 1.17 PJJ UPM / UPMET Chloroplasts are the organelles in plants and algae that carry out photosynthesis. It converts light energy into chemical energy in the form of ATP. Its structure has some resemblance to a mitochondrion. Within the chloroplasts are stacks of sac-like membrane structures known as thylakoids. 1.18 Cytosol is the aqueous part of the cytoplasm excluding the organelles. It contains many enzymes and is the main site for metabolism. It contains cytoskeleton which maintains the shape of the cell. It also contain storage granules such as glycogen (animal cell) and starch (plant cell). 1.19 Lysosomes and vacuoles are specific vesicles in cells. Lysosomes are found only in animal cells and contain enzymes that catalyse the breakdown of proteins and nucleic acids. Vacuoles are membrane bound vesicles containing fluid, normally found in mature plant cells. Vacuoles can have waste materials such as excess nitrogenous compounds. 1.20 Plant cell wall surrounds the plasma membrane. It is made of cellulose and polysaccharides with repeating glucose units. It provides protection against osmotic and mechanical rupture but yet porous to very small molecules. The cell wall gave strength to the cells and plant tissues. 8 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET TOPIC 2 : WATER, BONDING AND ITS INTERACTIONS Main Points 2.1 Water, H2O is the most abundant component of a cell and an indispensable compound for life. Some molecules are attracted to water and interact extensively with it whilst other molecules or part of it may avoid water molecules and can be partially or completely insoluble. 2.2 Water is just not only a universal solvent but also provides an aqueous environment for metabolic activity to function. It is an excellent solvent for both ionic substances (e.g. salts) and non-ionic substances (sugars, simple alcohols, amines, aldehydes and ketones). It is also a substrate for many cellular reactions. 2.3 The processes of life require a wide variety of ions and molecules to move about in proximity, that is, to be soluble in a common medium. Water serves as the universal intracellular and extracellular medium, due to its remarkable solvent ability. This ability is due to its dipolar (bipolar) nature and the tendency to form hydrogen bonds. 2.4 In a water molecule, the oxygen nucleus attracts electrons stronger than the single proton in the hydrogen nucleus, thus the oxygen is more electronegative than the hydrogen atom creating an uneven distribution of charges within each O-H bond. As a result oxygen will bear a partial negative charge (δ-) and hydrogen bears a partial positive charge (δ+). This unequal distribution of charge within a bond is known as dipole and the bond is said to be polar. 9 PRT3402- Agricultural Biochemistry 2.5 PJJ UPM / UPMET A polar bond is a covalent bond in which the electrons are shared unequally (e.g. H2O) whilst a non-polar bond is when the bondings electrons are equally shared (CH4). Polarity is governed by the polarity of its covalent bonds and geometry of structure. A water molecule is V-shaped with and angle of 104.5o between the two O-H bonds. This arrangement of the polar O-H bonds creates a permanent dipole. 2.6 Covalent bonds are the forces that hold atoms together as molecules e.g. the two O-H bonds in water molecule. Non-covalent interactions or non-covalent bonds are weak interactions between ions, molecules, and parts of molecules. They help shape individual molecules and groups of molecules and ions, but are weak enough to be continually broken and re-formed. Some of the most important covalent bonds in biology such as C-C and C-H have bond energies in the range of 300-400 kJ/mol. 10 PRT3402- Agricultural Biochemistry 2.7 PJJ UPM / UPMET Hydrogen bonding is the interaction of a hydrogen atom that is covalently bonded to one electronegative atom with a second electronegative atom. It can also be described as an interaction of a covalently bonded hydrogen atom on a donor group (-OH) and a pair of non-bonded electrons on an acceptor group, . The atom which is covalently bonded to hydrogen is the hydrogen bond donor and the atom with the non-bonded electron pair is the hydrogen bond acceptor. 2.8 Hydrogen bonds provide forces that help stabilize the structures of macromolecules, such as DNA and proteins, give a molecule like water its unusual chemical characteristics for its size and the hydrogen bonds of water also assist in solubilizing polar compounds. 2.9 The electron arrangement of a single water molecule is shown below. Two of the outer six electrons of the oxygen atom are involved in bonding to the hydrogens. The other four electrons exist in non-bonded pairs, which are excellent hydrogen bond acceptors. The OH groups in water are strong hydrogen bond donors. Each water molecule is simultaneously a hydrogen bond donor and a hydrogen bond acceptor, and a sample of water is a dynamic network of H-bonded molecules. Due to this the boiling point of water 11 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET (100oC) is high despite the relatively small molecular weight because of the strong tendency to form hydrogen bonds between the water molecules. 2.10 Substances that readily dissolve in water are hydrophilic (water-loving). Hydrophilic molecules are also ionic and engage in hydrogen bonding. They are either soluble in water or at least wettable. 2.11 The solubility of NaCl in water (hydration) is shown below. Each sodium ion (Na+) and chloride ion (Cl-) will be surrounded by water molecules. 12 PRT3402- Agricultural Biochemistry 2.13 PJJ UPM / UPMET Molecules that are readily soluble in water (e.g. sugar or glucose) have charged parts that are attracted to the charges within the water molecule. The presence of polar molecules will attract water molecules and the molecules, initially in crystal form will go into solution. It remains in solution because the glucose molecules are surrounded by water molecules. The group of water molecules surrounding another molecule is known as hydration shell. A glucose molecule in solution being surrounded by water molecules forming a hydration shell. 2.14 Hydrophobic (water-hating) molecules as opposed to hydrophilic substances are non-ionic and non-polar, not wettable and do not readily dissolve in water. They form a cage-like structure, whereby the water molecules ‘trapped’ the hydrophobic molecule. 13 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET A hydrophobic molecule ‘trapped’ in a cage-like structure of water molecules. 2.15 Other properties of water that takes advantage of the unique characteristics of water molecule and hydrogen bonding includes: a. a universal solvent b. occurs in three physical states – gas (steam), liquid and solid (ice) c. capillary action due to high surface tension d. absorb heat due to high specific heat index 2.15 Amphiphatic molecules or compounds have both hydrophobic and hydrophilic parts which are usually at opposite ends. Many biological compounds exhibited this property as indicated below. 14 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET 15 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET TOPIC 3 : ACID, BASE, pH and BUFFERS 3.1 The following table gives a definition of acid and base which are very important considerations in any biochemical reaction in living organisms. The biochemical behavior of many biological compounds depend on their acidbase properties since they have functional groups that react with (donate or accept) H+ ion. ACID BASE Definition Is a proton (H+) donor Is a proton acceptor, Properties Tastes sour (vinegar – Tastes bitter acetic acid, fruit juices- Turns litmus paper blue citric acid) Feel soapy Turns litmus paper red React with acids to form Concntrated acid burns salts skins React with acids to form salts Strong Examples: Examples: HCl - hydrochloric acid NaOH - sodium hydroxide H2SO4 - sulfuric acid KOH - potassium HNO3 - nitric acid hydroxide HClO4 - perchloric acid Ca(OH)2 - calcium hydroxide Strong acids ionize completely in water Strong bases completely forming H+ and an anion: disassociate in water into HCl (aq) → H+(aq) + Cl- the cations and OH-: (aq) NaOH(s) → Na+(aq) + OH-(aq) Weak hydrofluoric acid - HF Ammonia - NH3, acetic acid - CH3COOH. diethylamine (CH3CH2)2NH. 16 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET Partially dissociates in Ionize partially in water to water to give H+ and the OH- and its cations. anion. HF (aq) H+ (aq) + F- NH3 (aq) + H2O (aq) (aq) 3.2 NH4+ (aq) + OH- (aq) Acid and base will react in a neutralization reaction to form salt: Acid + base → salt + water (HCl (aq) + NaOH(aq) → NaCl(aq) + H2O(l) The following equation will give an indication of acidity or basicity of a solution. pH = - log10 [H+], [H+] = 1 x 10-x pH = x. 3.3 Substances are considered acidic, basic and neutral solutions based on the pH values. It is acidic if the pH is less than 7, basic if it is more than 7 and neutral if it is equal to 7. The ph scale is indicated below: 3.4 Water is amphoteric meaning that it can act as an acid or as a base. Water behaves as acid: NH3 + H2O base: HCl + H2O → base: H2O + H2O 3.5 H3O+ → NH4+ + OH-. Water acting as a + Cl-. Water acting as both acid and H3O+ + OH- The dissociation constant of water will show that pH of water is equivalent to 7. Kw = [H3O+] [OH-] = [H+][OH-] 17 PRT3402- Agricultural Biochemistry PJJ UPM / UPMET Kw = 1.0 x 10 -14 [H3O+] = [OH-] = 1.0 x 10 -7 3.6 Buffering refers to the ability of a solution to resist change in pH when acids or bases are added. A buffer solution contains a weak acid + its conjugate base or a weak base + its conjugate acid. H+ CH3COOH Acid conjugate base NH4+ NH3 + H2O Base 3.7 CH3COO- + + OH- conjugate acid The Henderson-Hasselbalch equation describes the chemical composition of a buffer as a function of pH. The equation is useful for estimating the pH of a buffer solution. [HA]=concentration of undisociated weak acid [A-]=concentration of the conjugate base [HA] pKa = -log10 Ka The Ka and pKa values of weak acids are useful when calculating the pH of buffer solutions. For e.g., what is the pH of a buffer solution containing 1M acetic acid and 0.5M sodium acetate? [Given that acetic acid: K a = 1.8 x 10-5, pKa = 4.7]. The answer can be calculated using the Henderson-Hasselbalch equation. The concentration of acetic acid (1M) corresponds to the [HA] term and the concentration of acetate (from sodium acetate) (0.5M) corresponds to the [A-] term. Hence, pH = 4.7 + log ([0.5]/[1.0]) = 4.4 3.8 In biological fluids phosphate (PO4) and carbonate (C03) acts as buffers. Other buffers such as TRIS and MES are used to buffer solutions at the appropriate pH in many biochemical laboratories. 18