Spectroscopy PP

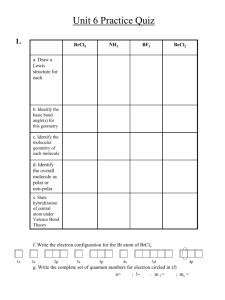
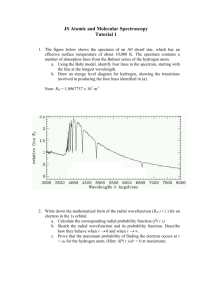
How does the wavelength of a light beam and the size of a slit it is going through control the amount of diffraction?
WORK
SLC
Expect more e-mails/phone calls if you’re not showing up
Physics Club
Missing Lens
Learn what spectrometry is and how scientists use it to identify specific atoms.
Be able to calculate the photons released from different energy level drops.
Let’s go over it.
What can electromagnetic waves tell us about stars, planets, and galaxies?
Spectroscopy is the process of obtaining a spectrum and reading the information it contains.
Each element has its own unique spectra.
If we collect the spectra of distant objects in our universe we can figure out what elements they are made of.
To perform spectroscopy you need a spectrometer
A spectrometer is an instrument used to measure properties of light over a specific portion of the electromagnetic spectrum
For visible light we will use a spectrometer that has a prism in it and we will use our eyes as the detector.
Different examples of spectrometers
Two ways
1. Draw what you see
2. Plot an Intensity vs.
Wavelength Graph
Continuous Spectrum
Spectrum of an ordinary light bulb; rainbow because it has all the visible wavelengths in it
Emission Line Spectrum
A thin cloud of gas emits light only at specific wavelengths that depend on its composition and temperature
Absorption Line Spectrum
If a cloud of gas is between us and a white light source, we still see most of the continuous light emitted by the light. However, the cloud absorbs light of specific wavelength and leaves dark lines
1.
What can a spectrometer tell us about a very distant object?
2.
What are the three types of spectra?
In 1913 Niels Bohr proposed that an atom has a positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus – similar to the structure of a solar system, but with the attraction provided by electrostatic forces rather than gravity.
Bohr said that when energy is added to an atom it becomes excited and the electrons can temporarily move up to higher orbits.
The Bohr Model says that as an electron returns to its normal orbit it releases the energy it previously absorbed in the form of a photon.
When incoming energy excites a hydrogen atom, its electron is moved into a higher energy level.
Atoms do not want to stay in higher energy levels
So as the electron returns to its original energy level it releases the energy that originally excited it as a photon (light particle)
The Bohr model has been superseded by quantum mechanics.
Electrons do not stay in perfect little orbits, nor are they held there by the electrostatic force.
Quantum mechanics says that electrons are in
“electron clouds” that show the probability of an electron being there.
1.
2.
What does an electron do as it absorbs energy?
What happens when an electron drops an energy level?
SLC
Expect more e-mails/phone calls if you’re not showing up
Physics Club
Missing Lens
Learn what spectrometry is and how scientists use it to identify specific atoms.
Be able to calculate the photons released from different energy level drops.
A specific photon is emitted during any energy level transition as long as the electron is dropping down at least one energy level.
To figure out the energy between any transition use:
This photon has a specific frequency that corresponds to the energy released from the atom
Remember:
We organize energy levels on “Energy Level
Diagrams”
Emission and absorption lines form as a direct consequence of the fact that each type of atom, ion, or molecule possesses a unique set of energy levels.
We know the energy levels of atoms, ions, and molecules so we just need to match our experimental observations with what we already know.
1electronvolt (eV)
= 1.60x10^-19 J
(Reference Table)
When an atom ionizes it loses all of its electrons
The negative eV value describes the difference in energy between an electron in an energy level and an electron infinitely far from the nucleus
Where
Z is the atomic number n is the energy level (1, 2, 3,….)
This only works as an approximation for a single electron (need quantum mechanics)
What is the energy of the photon emitted from a hydrogen atom when the electron falls from level 3 to 1?
Is this different than if it fell from level 3 to
2 and then 2 to 1?
n=3 to n=1;
n=3:-1.51eV
n=1:-13.60eV
𝐸 𝑝ℎ𝑜𝑡𝑜𝑛
= −1.51𝑒𝑉 − (−13.60𝑒𝑉)
𝐸
𝑝ℎ𝑜𝑡𝑜𝑛
= 12.09𝑒𝑉
n=3 to n=2
n=3:-1.51eV
n=2:-3.40eV
n=2 to n=1
n=2:-3.40eV
n=1:-13.60eV
𝐸
𝑝ℎ𝑜𝑡𝑜𝑛
= −3.40𝑒𝑉 − (−13.60𝑒𝑉)
𝐸
𝑝ℎ𝑜𝑡𝑜𝑛
= 10.20𝑒𝑉
What frequencies do the three different photons have?
𝐸
3 𝑡𝑜 1
= ℎ𝑓
1.6𝑥10
−19
𝐽
12.09𝑒𝑉
1𝑒𝑉 𝑓 = 2.9𝑥10
15
𝐻𝑧
𝐸
3 𝑡𝑜 2
= ℎ𝑓
1.6𝑥10
−19
𝐽
1.89𝑒𝑉
1𝑒𝑉 𝑓 = 4.6𝑥10
14
𝐻𝑧
= 6.63𝑥10 −34 𝐽 ∗ 𝑠(𝑓)
= 6.63𝑥10 −34 𝐽 ∗ 𝑠(𝑓)
𝐸
2 𝑡𝑜 1
= ℎ𝑓
1.6𝑥10
−19
𝐽
10.20𝑒𝑉
1𝑒𝑉 𝑓 = 2.4𝑥10 15 𝐻𝑧
= 6.63𝑥10 −34 𝐽 ∗ 𝑠(𝑓)
Would we be able to see all three photons with our eyes?
No, only the photon released from energy level 3 to 2 falls within the visible spectrum of the EM radiation.
Page 772 answer questions 29, 30, 32, 33
29.
30.
31.
32.
33.
3.91eV; this energy corresponds to level E6
1.91eV
Skip it.
A) 2.72eV B) 3.06eV
1.24eV; 2.99x10^14Hz
Regents Part 2
June 2014
Due Friday: Even if you miss class
Using a spectrometer with LEDs demo
Why aren’t the colors in very thin circles?
What can a spectrometer tell you about how a fluorescent light works?
Missing Lens
Homework due Friday
No Physics Club today
Tomorrow
SLC Thursday
We will use 5 samples
Draw what you see
Compare it to a known spectrum
Record spectra on a separate sheet of graph paper. Your scale should range from 400nm to 700nm.
Use the whole width of the paper.
400nm
450nm
500nm
550nm
600nm
650nm
700nm