Electron Domain
advertisement

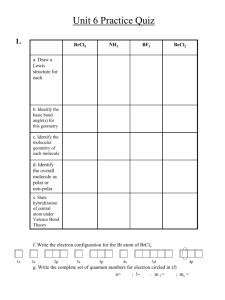
Shapes of molecules (molecules actually have 3D structures that affect how they bond in reactions) Based on electrons and their repulsion from each other Valence Shell Electron Pair Repulsion (VSEPR) Focused on general model ABx A = central atom, B = terminal (attached) atoms Recall: Electron Pairs can be bonding pairs or lone pairs Bonding pairs may be single bonds or multiple bonds An electron domain is a lone pair or a bonded pair (double and triple bonds only count for ONE electron domain) How many electron domains do these molecules have? Geometric shapes based on electron domains AB2 Linear 180° (provided no lone pairs on central) Ex: CO2 AB3 Trigonal Planar 120° (provided no lone pairs on central) Ex: BF3 Geometric shapes based on electron domains AB4 Tetrahedral 109.5° (provided no lone pairs on central) Ex: CH4 AB5 Trigonal Bipyramidal 120° (equatorial atoms) & 90° (axial atoms) Ex: PCl5 Geometric shapes based on electron domains AB6 Octahedral 90° between all atoms (provided no lone pairs on central) Ex: SF6 It is helpful to draw molecules on paper with 3-Dimensionality to better visualize the shape Electron Domain: based on electron domains (bonded and unbonded) Basically, it looks at ALL the electrons Molecular Geometry: based on atoms ONLY (bonded pairs only) Basically only looks at electrons in bonds (or atoms) 1) Draw Lewis Structure 2) Count electron domains around central atom 3) Determine Electron Domain Geometry 4) Determine Molecular Geometry from atoms only Consider the following three compounds: CH4, NH3, H2O What are their electron domain geometries? What are their molecular geometries? Some electron domains repel MORE than others Lone pairs repel more Because not bonded, they experience less nuclear attraction Double and Triple bonds repel more Due to higher electron density Other examples Determine the electron domain geometry and the molecular geometry for the following. Include bond angles as well. SeO2 SF6 State the electron domain geometry, the molecular geometry and the bond angle (if the bond angle is not ideal, state “less than _____°”) PCl3 SeCl4 A central atom is one where it is surrounded by 2 or more atoms Determine the multiple shapes in the molecule around each central atom Ex: Recall: A bond between 2 atoms is polar covalent when difference in electronegativity is greater than 0.5 A molecule OVERALL can also be polar or nonpolar Based on geometry of molecule and Individual bonds in molecule CO2 and H2O First, check if there are lone pairs of electrons If yes, it is POLAR (unless they are “balanced”) If no lone pairs, check if there are polar bonds If there are no polar bonds, is is NON POLAR If there are polar bonds, check for symmetry If all terminal atoms are the same, it is NON POLAR If terminal atoms are not the same, but there is symmetry, it is NON POLAR If terminal atoms are not the same, and there is no symmetry, it is POLAR Determine if the molecule is POLAR or NON POLAR CH2Cl2 For each hypothetical molecule below, draw one structure that is polar and one that is non polar H2SF4 & & XeF4 H2PCl3 For each molecule below, determine the electron domain geometry, the molecular geometry, draw the 3D-structure on paper, state if it is polar or nonpolar SbCl5 I 3- Definition: atoms share electrons when an atomic orbital on one atom overlaps with an atomic orbital on the other Explains why atoms share electrons Potential energy of resulting molecule is lower than combined energies of isolated atoms A.K.A. when bonds form, energy given off Therefore, energy required to break bonds Consider BeCl2 How can it bond with 2 atoms??? Hybridization: mixing of atomic orbitals Hybrid orbitals are always shown in GREEN Consider BF3 Consider CH4 Consider PCl5 Consider SF6 1) 2) 3) 4) 5) Draw Lewis Structure Count # of electron domains (this is # of hybrid orbitals needed) Determine the hybrid orbital needed Draw ground state configuration of central atom Place electrons in the hybrid orbitals one at a time Use hybrid orbital theory to explain the bonding in BrF5 Show the hybridization of the following molecule. Be sure to include a) what type of hybrid orbital is needed and 2) the orbital diagram of the hybrid orbital XeF4 How many electron domains around the carbons? What hybrid orbitals are needed? Bonds that are made using hybrid orbitals (or along the internuclear axis) are called sigma (σ) bonds Bonds that are made using remaining p orbitals and overlap on the sides are called pi (π) bonds