Alcohols, Phenols,
Thiols, and Ethers
Chapter 13
Introduction
• Alcohol – organic compound that contains a hydroxyl (-OH) group attached to an alkyl group
• Phenol – organic compound that contains a hydroxyl (-OH) group attached to an aryl group
• Ether – organic compound that has two alkyl or aryl groups attached to the oxygen atom; can be thought of as a substituted alcohol.
Examples
Alcohols -OH hydroxyl CH
3
-OH
CH
3
CH
2
-OH
OH
OH
Phenols
Ethers -OCH
3
-O-CH
3
Practice Problem 1
Classify each as an alcohol (1), phenol (2), or an ether (3):
A. _____ CH
3
CH
2
-O-CH
3
OH
B. _____
C. _____ CH
3
CH
2
OH
CH
3
Solution 1
Classify each as an alcohol (1), phenol (2), or an ether (3):
A. ____ CH
3
CH
2
-O-CH
3
OH
B. _ __
CH
3
C. ____ CH
3
CH
2
OH
Structure and Properties
• R-O-H portion of alcohol is similar to the structure of water.
– The oxygen and two atoms bonded to it lie in the same plane.
– The bond angle is 104°
• Hydroxyl groups are very polar because of significantly different electronegativities.
– Hydrogen bonding can form between alcohol molecules.
Hydrogen Bonding
R
O
H
O
H
O
H
R
H
H
O
H
Results of Hydrogen Bonding
• Alcohols boil at much higher temperatures than hydrocarbons of similar molecular weight.
• Alcohols with fewer than five carbons are very soluble in water.
• Alcohols with five to eight carbons are moderately soluble in water.
• As the nonpolar (R) portion of the alcohol gets larger, the water solubility decreases.
Solubility of Alcohols
• Very large alcohols are not soluble in water.
• Hydrophobic – “water fearing”; used to describe nonpolar region of molecule
• Hydrophilic – “water loving”; used to describe polar region of molecule
CH
3
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
OH
Hydrophobic
Hydrophilic
Solubility of Alcohols
• An increase in the number of hydroxyl groups will increase the influence of the polar hydroxyl group.
– Diols and triols are more water soluble than alcohols with only a single hydroxyl group.
Practice Problem 2
• Circle the more soluble alcohol in each pair.
A.
H
3
C C
H
2
OH
OR
H
3
C
H
2
C
C
H
2
H
2
C
OH
OH
OR
B.
OH
OH
OH
C.
OR
Solution 2
• Circle the more soluble alcohol in each pair.
OH
H
2
C
H
2
C
A.
H
3
C C
H
2
OR
H
3
C C
H
2
OH
OH
OR
B.
OH
OH
OH
C.
OR
Nomenclature of Alcohols
• A carbon compound that contain -OH
(hydroxyl) group
• In IUPAC name, the -e in alkane name is replaced with -ol.
CH
4 methane
CH
3
OH
CH
3
CH
3 methanol (methyl alcohol) ethane
CH
3
CH
2
OH ethanol (ethyl alcohol )
More Names of Alcohols
• IUPAC names for longer chains number the chain from the end nearest the -OH group.
CH
3
CH
2
CH
2
OH
OH
CH
3
CHCH
3
CH
3
OH
CH
3
CHCH
2
CH
2
CHCH
3
Alcohols that contain more than one hydroxyl group
• Alcohols containing two hydroxyl groups are named –diols.
• Alcohols containing three hydroxyl groups are named –triols.
• A number giving the position of each of the hydroxyl groups is needed in these cases.
Practice Problem 3
Name the following alcohols:
A. OH
B.
CH
3
CHCHCH
2
CH
3
CH
3
OH
Solution 3
Name the following alcohols:
A. OH
B.
CH
3
CHCHCH
2
CH
3
CH
3
OH
Homework
Pages 379-380 13.11-
13.14
Page 360 13.1 – 13.2
Due Tomorrow!!!!
Section 13.3
• It is your responsibility to read over section 13.3 – Medically
Important Alcohols (pgs. 360-
361) and take notes on the material covered!
• It will be on the weekly quiz and the test!
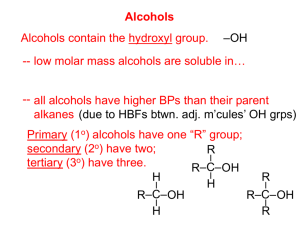
Classification of Alcohols
H
• Alcohols can be classified as primary (1°), secondary (2 °), or tertiary (3°) depending on the number of alkyl groups attached to the carbon bearing the –OH group.
H H CH
3
CH
3
C H H
3
C C OH H
3
C C H H
3
C C CH
3
OH H OH OH
Methanol 1° alcohol 2° alcohol 3° alcohol ethanol 2-propanol 2-methyl-2-propanol
Practice Problem 4
Classify each of the following alcohols as primary, secondary, tertiary, or aromatic.
1. 1-butanol
2. 3-pentanol
3. 1-methylcyclopentanol
4. 2-methyl-2-pentanol
1. 1-butanol
Solution 4
2. 3-pentanol
3. 1-methylcyclopentanol
4. 2-methyl-2-pentanol
Homework
• Pages 380-381: 13.15,
13.16, 13.18, 13.19, 13.20,
13.21, 13.22, 13.27, and
13.28
Due Tomorrow!
Preparation of Alcohols
• REVIEW: Hydration – an addition reaction in which a water molecule is added to an alkene; requires acid as catalyst
R
R R
H
H+
+
R H
O H
R OH
R R
Alkene Water
R
Alcohol
Markovnikov’s rule applies!!
• Ethene
Hydration Examples
• 1-butene
Preparation of Alcohols
• Hydrogenation of aldehydes and ketones
OH
O
+
H
Catalyst
R
1
R
2
H
R
1
R
2
H
In an aldehyde, R1 and R2 may be either alkyl groups or H.
In a ketone, R1 and R2 are both alkyl groups.
We will discuss this in more detail in section 14.4.
Hydrogenation Examples
• Ethanal
• 2-propanone
Preparation of Alcohols
YOU TRY THESE!
+
Dehydration of Alcohols
• Alcohols undergo dehydration (lose water) when heating with concentrated sulfuric or phosphoric acid.
• Dehydration is an example of an elimination reaction.
• Elimination reaction – a reaction in which a molecule loses atoms or ions from its structure.
• Dehydration is opposite of hydration!
Dehydration of Alcohols
• General Reaction
H
+
RCH
2
CH
2
OH
Heat
RCH=CH
2
+ H
2
O
• Ethanol
H
+
CH
3
CH
2
OH
Heat
• 2-butanol
H
H H OH H
C C C C
H H H H
H
H
+
Heat
Dehydration of Alcohols
• Zaitsev’s rule – in an elimination reaction, the alkene with the greatest number of alkyl groups on the double bonded carbon
(more highly substituted alkene) is the major product of the reaction
• Another example:
Oxidation Reactions
• Oxidation – loss of electrons; add O and/or lose H
• Reduction – gain of electrons; add H and/or lose O
• Common oxidizing agents:
– Basic potassium permanganate (KMnO
4
/OH )
– Chromic Acid (H
2
CrO
4
)
• [O] Any general oxidizing agent
Oxidation of Primary Alcohols
• General equation:
– Primary alcohol aldehyde
RCH
2
CH=O
EXAMPLE:
1-propanol
Oxidation of Secondary
Alcohols
• General equation:
– Secondary alcohol ketone
O
R-C-R
EXAMPLE:
2-propanol
Oxidation of Tertiary Alcohols
• General equation:
– Tertiary alcohol NO REACTION!!!
H
H
H H OH
YOU TRY THESE!
H
[O]
C C C C H
H H H
[O]
OH
[O]
H O
CH
3
CH
2
OH
[O]
Phenols
• Phenols – compounds in which the hydroxyl group is attached to a benzene ring
• They are polar compounds because of the polar hydroxyl group.
• Smaller phenols are somewhat soluble in water.
• They are found in fragrances and flavorings and are also used as preservatives and germicides.
Common Phenols and their
Uses
(CH
3
)
2
HC CH
3
Thymol (mint)
(CH
3
)
2
HC
OH
OH
CH
3
Carvacrol (savory
)
Common Phenols and their
Uses
C(CH
3
)
3
CH
3
OH
OH
Butylated hydroxytoluene,
BHT(Food preservative)
(CH
3
)
3
C
Phenol
(Carbolic acid when dissolved in water)
Antiseptic and disinfectant used by
Joseph Lister to bathe wounds and sterlize instruments
Ethers
Ethers
• What are they?
• How are they used?
• What do you know about them?
Structure of Ethers
• Similar structure to alcohols
Alcohols: R-OH
–OH is “ hydroxy” group
Ethers: R-OR
–OR is “ alkoxy” group
Alkoxy group
• “R” groups can be the same:
• “R” groups can be different:
CH
3
-O-CH
3
• Both “R” groups have one carbon
CH
3
CH
2
CH
2
-O-CH
3
• One “R” group has three carbons while the other has one
Alkoxy group
• The root names are used with –oxy to name that portion of the ether:
– meth- + -oxy
– eth- + -oxy = methoxy
= propoxy
Naming ethers – IUPAC Way
1.
Find the root name of the smaller “R” group
Ex: meth-, eth-, prop-
2. Add –oxy
Ex: methoxy, ethoxy, propoxy
3.
Add the full name of the larger “R” group
EXAMPLE:
O methoxypropane
Name that ether
O
O
O
Draw the structure of:
1. methoxypropane
2. methoxyoctane
3. propoxypropane
4. ethoxypentane
Naming Ethers – Common
Name
• Name by placing the names of the two alkyl groups attached to the ether oxygen as words in front of the word “ether.” Typically this is done in alphabetical order.
O
Diethyl Ether
O
O
Ethyl methyl ether
Butyl propyl ether
Properties of ethers
• The C-O bond in ethers is polar, making the molecule polar
• Ethers have very low boiling points
• Ethers are relatively inert, but flammable in air
Let’s compare…
CH
3
Ether
–O-CH
2
CH
3
Molecular Formula:
C
3
H
8
O
Molecular Weight:
60.09g/mol
Boiling Point:
7.9
° C
Alcohol
CH
3
CH
2
CH
2
-OH
Molecular Formula:
C
3
H
8
O
Molecular Weight:
60.09g/mol
Boiling Point:
97.2
° C
Preparing ethers
• Ethers are formed from alcohols
ROH + R’-OH
H
R-OR’ +H
2
O
– R and R’ can be the same or different
– Called a dehydration reaction
Predict the products
CH
3
OH + CH
3
CH
2
OH
H+
CH
3
CH
2
CH
2
OH + CH
3
CHCH
3
H+
OH
Common uses
• Ethers as anesthetics:
– Penthrane
– Enthrane
• Ethers as additives in gasoline
– MTBE – methyl tert-butyl ether
Thiols
• Compounds that contain the sulfhydryl group (-SH)
• Similar to alcohols in structure, but the sulfur atom replaces the oxygen atom
• Have nauseating aromas – defense spray of North American striped skunk
H CH
3
Trans-2-butene-1-thiol
H
2
C H
SH
Naming Thiols
• Use the same rules as for alcohols except that the full name of the alkane is retained.
• Add the suffix –thiol.
CH
3
CH
2
SH
CH
3 ethanethiol
CH
3
CHCH
2
CH
2
3-methyl-1-butanethiol
SH
HSCH
2
CH
2
SH
1,2-ethanedithiol
Uses of Thiols
• Thiols are involved in protein structure and conformation.
• Cysteine is an amino acid that contains a sulfhydryl group.
• BAL (British Anti-Lewisite) is used as an antidote for mercury poisoning.
• Coenzyme A serves as a carrier of acetyl groups in biochemical reactions.
Homework
•Pg. 381-382: 13.43,
13.44, 13.45, 13.48,
13.49, 13.51, 13.52,
13.53, 13.59, 13.60
 0
0