Closed System Energy Balance Examples
advertisement

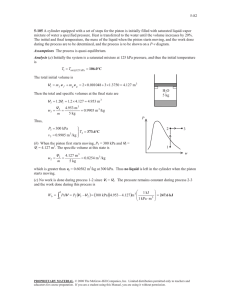
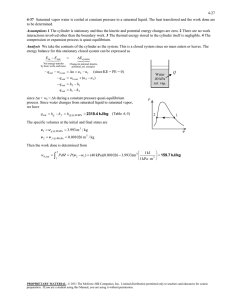
Closed System Energy Balance Example 5.0 kg of water is heated from 110oC to 400oC at a constant pressure of 200 kPa. Determine the amount of heat transfer. Closed System Energy Balance Example A freely moving piston-cylinder device contains hydrogen at 1000 kPa and 50°C. The initial volume of the system is 2 L and it is heated until the volume reaches 10 L. Determine the amount and direction of heat transfer, in kJ. Closed System Energy Balance Example A freely moving piston cylinder device initially contains 3 kg saturated liquid water at 150C. An electric resistance heater is placed in the tank and is turned on for 15 min until the volume triples. Determine… a. The initial volume of the tank b. The final temperature c. The electrical power of the heater, in kW Closed System Energy Balance Example A student living in a 4 m x 6 m x 6 m dorm room turns on a 150 W fan before leaving for 10 hours on a hot summer day. Assume that all windows and doors are well sealed and the that the walls are adiabatic. When the student left, the temperature of the room was 25oC and the pressure was 100 kPa. Determine the temperature when the student returns. Energy Balance Example The power goes out at your brewery due to an overheated transformer, shutting down your fermentation cooling mechanism. Consider a 25 m3 cylindroconical vessel that is full with a product at 10oC, specific heat of 3.4 kJ/kg.K, and density of 1025 kg/m3. Assuming that the sum of heat gains from the surroundings and conversion from fermentation is 4 kW, determine the temperature after 4 hours. How would the 4 kW load change over time? Closed System Energy Balance Example Two tanks are separated by a partition. Initially, one tank contained 2 kg of steam at 1 Mpa and 300oC while the other contained 3 kg of a saturated mixture (water) at 150oC with a quality of 50%. The partition is removed, the two sides mix and reach equilibrium. The final pressure of the mixture is 300 kPa. Determine the temperature and quality of the steam (if mix) at the final state and the amount of heat loss from the tanks. Closed System Energy Balance Example A mass of 5 kg of saturated liquid-vapor mixture water is contained in a piston-cylinder device at 125 kPa. Initially, 2 kg is in liquid phase. Heat is transferred to the system until the pressure reaches 300 kPa, at which time the piston rises from a set of stops. Heat transfer increases until the volume increases by 20%. Determine the initial and final temperatures, the mass of liquid when the piston starts moving and the work done. Plot on a P-v.