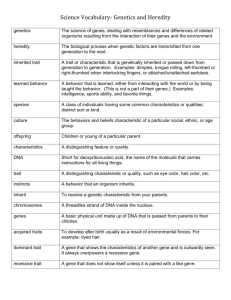
marker-assisted selection (mas)

THE USE OF
GENETIC MARKERS
IN PLANT BREEDING
Use of Molecular Markers
Clonal identity,
Family structure,
Population structure,
Phylogeny (Genetic Diversity)
Mapping
Parental analysis,
Gene flow,
Hybridisation
Genetic Diversity
Define appropriate geographical scales for monitoring and management (epidemology)
Establish gene flow mechanism
Identify the origin of individual (mutation detection)
Monitor the effect of management practices
Manage small number of individual in ex situ collection
Establish of identity in cultivar and clones (fingerprint)
Paternity analysis and forensic
Genetic Diversity
Clonal Identity
seeds, plantlets fingerprints
early selection of the good allele
Mapping
The determination of the position and relative distances of gene on chromosome by means of their linkage
Genetic map
A linear arrangement of genes or genetic markers obtained based on recombination
Physical map
A linear order of genes or DNA fragments
Physical Mapping
It contains ordered overlapping cloned DNA fragment
The cloned DNA fragments are usually obtained using restriction enzyme digestion
Genetic Maps
Molecular markers (especially RFLPs and SSRs) can be used to produce genetic maps because they represent an almost unlimited number of alleles that can be followed in progeny of crosses.
Chromosomes with morphological marker alleles
T
R t r
Chromosomes with molecular marker alleles
RFLP1b
RFLP2b
SSR1b
RFLP1a
RFLP2a
SSR1a
RFLP3b
RFLP3a
SSR2b
SSR2a
RFLP4b
RFLP4a or
QTL (Quantitative Trait Loci)
A locus or DNA segment that carries more genes coding for an agronomic or other traits
Individual loci responsible for quantitative genetic variation
Region in the genome containing factors influencing a quantitative trait
Region identified by statistical association
QTL Mapping
A set of procedures for detecting genes controlling quantitative traits (QTL) and estimating their genetics effects and location
Localizing and determining a segment of DNA that regulate quantitative traits
Detecting and locating gene having an effect on a quantitative traits
To assist selection
Marker Assisted Selection
Types of traits
Single gene trait: seed shape Multigenic trait; ex: plant growth
=Quantitative Trait Loci
Linkage groups
Developing a Marker
Best marker is DNA sequence responsible for phenotype i.e. gene
If you know the gene responsible and has been isolated, compare sequence of wild-type and mutant DNA
Develop specific primers to gene that will distinguish the two forms
Developing a Marker
If gene is unknown, screen contrasting populations
Use populations rather than individuals
Need to “blend” genetic differences between individual other than trait of interest
Developing Markers
Cross individual differing in trait you wish to develop a marker
Collect progeny and self or polycross the progeny
Collect and select the F2 generation for the trait you are interested in
Select 5 - 10 individuals in the F2 showing each trait
Developing Markers
Extract DNA from selected F2s
Pool equal amounts of DNA from each individual into two samples - one for each trait
Screen pooled or “bulked” DNA with what method of marker method you wish to use
Conduct linkage analysis to develop QTL Marker
Other methods to develop population for markers exist but are more expensive and slower to develop
→ Near Isogenic Lines, Recombinant Inbreeds, Single
Seed Decent
MAS
Marker assisted selection
The use of DNA markers that are tightly-linked to target loci as a substitute for or to assist phenotypic screening
Assumption
DNA markers can reliably predict phenotype
Marker Assisted Selection
Breeding for specific traits in plants is expensive and time consuming
The progeny often need to reach maturity before a determination of the success of the cross can be made
The greater the complexity of the trait, the more time and effort needed to achieve a desirable result
The goal to MAS is to reduce the time needed to determine if the progeny have trait
The second goal is to reduce costs associated with screening for traits
If you can detect the distinguishing trait at the DNA level you can identify positive selection very early.
CONVENTIONAL PLANT BREEDING
Recipient
P
1 x
P
2
Donor
F
1 large populations consisting of thousands of plants
F
2
PHENOTYPIC SELECTION
Salinity screening in phytotron
Glasshouse trials
Bacterial blight screening
Phosphorus deficiency plot
Field trials
MARKER-ASSISTED BREEDING
Susceptible
P
1 x P
2
Resistant
F
1
F
2 large populations consisting of thousands of plants
MARKER-ASSISTED SELECTION (MAS)
Method whereby phenotypic selection is based on DNA markers
Advantages of MAS
Simpler method compared to phenotypic screening
• Especially for traits with laborious screening
• May save time and resources
Selection at seedling stage
• Important for traits such as grain quality
• Can select before transplanting in rice
Increased reliability
• No environmental effects
• Can discriminate between homozygotes and heterozygotes and select single plants
Potential benefits from MAS
more accurate and efficient selection of specific genotypes
• May lead to accelerated variety development more efficient use of resources
• Especially field trials
Crossing house
Backcross nursery
Overview of
‘marker genotyping’
(1) LEAF TISSUE
SAMPLING
(2) DNA EXTRACTION
(3) PCR
(4) GEL ELECTROPHORESIS
(5) MARKER ANALYSIS
Developing a Marker
Best marker is DNA sequence responsible for phenotype i.e. gene
If you know the gene responsible and has been isolated, compare sequence of wild-type and mutant DNA
Develop specific primers to gene that will distinguish the two forms
Developing a Marker
If gene is unknown, screen contrasting populations
Use populations rather than individuals
Need to “blend” genetic differences between individual other than trait of interest
Developing Markers
Cross individual differing in trait you wish to develop a marker
Collect progeny and self or polycross the progeny
Collect and select the F2 generation for the trait you are interested in
Select 5 - 10 individuals in the F2 showing each trait
Developing Markers
Extract DNA from selected F2s
Pool equal amounts of DNA from each individual into two samples - one for each trait
Screen pooled or “bulked” DNA with what method of marker method you wish to use
Conduct linkage analysis to develop QTL Marker
Other methods to develop population for markers exist but are more expensive and slower to develop
→ Near Isogenic Lines, Recombinant Inbreeds, Single
Seed Decent
Considerations for using DNA markers in plant breeding
Technical methodology
• simple or complicated?
Reliability
Degree of polymorphism
DNA quality and quantity required
Cost**
Available resources
• Equipment, technical expertise
Markers must be tightly-linked to target loci!
Ideally markers should be <5 cM from a gene or QTL
RELIABILITY FOR
SELECTION
Marker A
5 cM
QTL Using marker A only:
1 – r
A
= ~95%
Marker A
Marker B
5 cM
QTL
5 cM
Using markers A and B:
1 - 2 r
A r
B
= ~99.5%
• Using a pair of flanking markers can greatly improve reliability but increases time and cost
Markers must be polymorphic
RM84
1 2 3 4 5 6
7 8
RM296
1 2 3 4 5 6 7
8
P
1
P
2
P
1
P
2
Not polymorphic Polymorphic!
DNA extractions
Mortar and pestles
Porcelain grinding plates
LEAF SAMPLING
Wheat seedling tissue sampling in Southern Queensland,
Australia.
High throughput DNA extractions “Geno-
Grinder”
PCR-based DNA markers
Generated by using Polymerase Chain Reaction
Preferred markers due to technical simplicity and cost
PCR Buffer +
MgCl
2
+ dNTPS +
Taq +
Primers +
DNA template
PCR
THERMAL CYCLING
GEL ELECTROPHORESIS
Agarose or Acrylamide gels
Marker Assisted Selection
Useful when the gene(s) of interest is difficult to select:
1. Recessive Genes
2. Multiple Genes for Disease Resistance
3. Quantitative traits
4. Large genotype x environment interaction
MARKER ASSISTED
BREEDING SCHEMES
3.
4.
1.
2.
Marker-assisted backcrossing
Pyramiding
Early generation selection
‘Combined’ approaches
Marker-assisted backcrossing
(MAB)
MAB has several advantages over conventional backcrossing:
• Effective selection of target loci
• Minimize linkage drag
• Accelerated recovery of recurrent parent
1 2 3 4
1 2 3 4
Target locus
1 2 3 4
TARGET LOCUS
SELECTION
RECOMBINANT
SELECTION
BACKGROUND
SELECTION
FOREGROUND
SELECTION
BACKGROUND SELECTION
Gene Pyramiding
Widely used for combining multiple disease resistance genes for specific races of a pathogen
Pyramiding is extremely difficult to achieve using conventional methods
Consider: phenotyping a single plant for multiple forms of seedling resistance – almost impossible
Important to develop ‘durable’ disease resistance against different races
Process of combining several genes, usually from 2 different parents, together into a single genotype
Breeding plan
P
1
Gene A x P
1
Gene B
F
1
Gene A + B
Genotypes
P
1
: AAbb x P
2
: aaBB
F
1
: AaBb
F
2
MAS
Select F2 plants that have
Gene A and Gene B
F
2
AB
Ab aB ab
AB Ab aB ab
AABB AABb AaBB AaBb
AABb AAbb AaBb Aabb
AaBB AaBb aaBB aaBb
AaBb Aabb aaBb aabb
Early generation MAS
MAS conducted at F2 or F3 stage
Plants with desirable genes/QTLs are selected and alleles can be ‘fixed’ in the homozygous state
• plants with undesirable gene combinations can be discarded
Advantage for later stages of breeding program because resources can be used to focus on fewer lines
Susceptible
P
1
F
1
F
2 x P
2
Resistant large populations (e.g. 2000 plants)
MAS for 1 QTL – 75% elimination of (3/4) unwanted genotypes
MAS for 2 QTLs – 94% elimination of (15/16) unwanted genotypes
PEDIGREE METHOD
P1 x P2
F1
SINGLE-LARGE SCALE MARKER-
ASSISTED SELECTION (SLS-MAS)
P1 x P2
F1
F2
Phenotypic screening
F2 MAS
F3
F4
F5
Plants spaceplanted in rows for individual plant selection
Families grown in progeny rows for selection.
F3
F4
F5
Only desirable
F3 lines planted in field
Families grown in progeny rows for selection.
Pedigree selection based on local needs
F6
Preliminary yield trials. Select single plants.
F6
F7
Further yield trials
F8 – F12
Multi-location testing, licensing, seed increase and cultivar release
F7
F8 – F12
Multi-location testing, licensing, seed increase and cultivar release
Benefits: breeding program can be efficiently scaled down to focus on fewer lines
Combined approaches
In some cases, a combination of phenotypic screening and MAS approach may be useful
1. To maximize genetic gain (when some QTLs have been unidentified from QTL mapping)
2. Level of recombination between marker and QTL
(in other words marker is not 100% accurate)
3. To reduce population sizes for traits where marker genotyping is cheaper or easier than phenotypic screening
‘
Marker-directed’ phenotyping
(Also called ‘tandem selection’)
Recurrent
Parent
P
1
(S) x P
2
(R)
F
1
(R) x P
1
(S)
Donor
Parent
BC
1
F
1 phenotypes: R and S
Use when markers are not
100% accurate or when phenotypic screening is more expensive compared to marker genotyping
MARKER-ASSISTED SELECTION (MAS)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 …
PHENOTYPIC SELECTION
SAVE TIME & REDUCE
COSTS
*Especially for quality traits*