File
advertisement

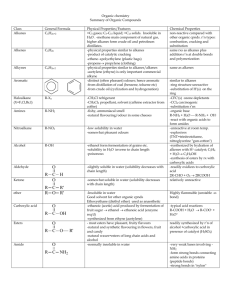
Carboxylic Acids (alkanoic acids) • Carboxylic acids contain a carbonyl group with an -OH attached. • The carboxyl functional group is -COOH: O R OH • Carboxylic acids are weak acids. • Named like alkanes with “-oic acid” at the end. • Typical carboxylic acids are found in spinach, vinegar, cleaners, vitamin C, aspirin, and citrus fruits. • Carboxylic acids are also used to make polymers for fibers, paints, and films. Reactions of Carboxylic Acids 1. Donate proton to a base (form alkanoate ion) 2. Esterification (squeeze play) – react with alcohol to form an ester. Also referred to as condensation. O O R 3. OH + R' HO R' R + H2O O 3. Amide formation (also condensation) – react with amine to form an amide. O O H R OH + R' N H R' R N H + H2O Esters • Some common esters are: benzocaine (in sun burn lotions), ethyl acetate (nail polish remover), vegetable oils, polyester thread, and aspirin. O • Esters contain -COOR groups: R OR' • Esters can be prepared by reacting a carboxylic acid with an alcohol and eliminating water: O O + H2O HO CH CH + 2 3 C C H3C OCH2CH3 H3C OH • Esters are named first using the alcohol part and then the acid part (in the above example: ethyl from ethanol and ethanoate from ethanoic acid). • Esters tend to have characteristic odors and are used as food flavorings and scents. Amines • Amines are organic bases. • Just as alcohols can be thought of organic forms of water, amines can be thought of organic forms of ammonia. • Treat NH2 groups as substituents called “amino” Examples 1. 1-aminobutane NH 2 2. NH2 NH2 1,6-diaminohexane Amides • Amides are composites of carbonyl and amine O functionalities: C R N R' H • Named by longest chain followed by “anamide” NH 2 Example: propanamide O Nitriles • Contain a cyanide group (CN) • Named as alkane (full name) followed by “nitrile” C N Example: butanenitrile • Halogenoalkanes (chloroalkanes, bromoalkanes) – contain a halogen atom in place of a hydrogen. Halides are treated as substituents Examples 1. 3-chlorohexane Cl 2. 3. Br 2,3-dibromohexane Br Br 2-bromo-2-chloropropane Cl Reactions of halogenoalkanes – nucleophilic substitution the “X” atom is replaced by a “nucleophile.” • Nucleophile – something with an unshared pair of electrons (attracted to a partially-positive carbon atom) • Examples: OH-, NH3, CN-, R’-O, R’-NH2, H2O, etc. • Hydroxide (OH-) is a better nucleophile than water (H2O), since it is more attracted to the partially-positive carbon atom. Primary halogenoalkanes (contain only 1 carbon attached to the carbon with the halogen atom) – react by a mechanism called SN2 (heterolytic fission of the bond): S = Substitution N = Nucleophilic 2= bimolecular H R Nu: C H H X Nu C R + X- H Curvy (curly) arrows indicate the movement of electrons. Rate of SN2 depends on: – concentrations of both reactants – strength of nucleophile (CN- > OH- > NH3 > H2O) – identity of halogen (bond strength) • I is fastest and F is slowest Tertiary halogenoalkane – SN1 reaction (heterolytic fission) – faster than SN1 1 = unimolecular (RDS) – rate depends only on the concentration of the halogenoalkane R 1. R' C R X R' R" C+ + X- R" Carbocation R R 2. R' C+ R" :Nu R' C R" Nu Examples – Write reactions for these nucleophilic substitutions, using “curly arrows.” 1. Potassium cyanide + H H C C3H7 2. Ammonia + Cl Br Elimination Reactions of Bromoalkanes • Elimination of HBr molecule to form an alkene. Occurs in NaOH in hot ethanol, heated under reflux. The H and Br are removed from neighboring carbon atoms. • E1 – unimolecular elimination reaction – a carbocation is formed (similar to SN1), then the OH- removes the H: H H H H C C H Br H H C C H OH- H H H H C C H H H H H H C C H