AP Bio Lab 1 Water Potential
advertisement

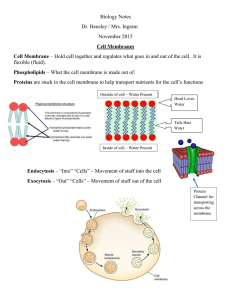
Water Potential The combined effect of: Solute concentration Pressure …are incorporated into a single measurement called water potential Water moves from the area of HIGH water potential to the area of LOW water potential when moving across a membrane *H20 moves from areas of high pressure to areas of low pressure* Solute Potential Pure water has a solute potential of zero. Solute potential is NEVER positive. Adding more solute is a negative experience; the solute potential always becomes negative or more negative Sample Problem The molar concentration of a sugar solution in an open beaker has been determined to be 0.3M. Calculate the solute potential at 27 degrees. Round your answer to the nearest hundredth. If the sugar solution was placed inside of dialysis tubing and put in a beaker of pure water (Ψ=1) what would happen? Why? Pressure Potential Is the sum of all pressure on water Get b/c of 2 things: Turgor pressure- force created by the cell membrane pushing against the cell wall (must have water inside to push cell wall out) You CAN’T know this unless given so assume that the pressure potential is zero unless otherwise told QUIZ TIME!!!! Lab Set Up Changes to procedure: Part B, bags are 15cm long NOT 30cm Part B, add 10ml of each sucrose solution to each bag You don’t know the molarities they are labeled (A-F). You must determine which solution goes to which molarity based on your collected data. Put all dialysis bags and string in the same beaker for the weekend and cover! No cork borer for potato cylinders so cut 4 relatively equally sized cubes for each solution REMOVE ALL SKIN (set up scales first before getting so you can mass and drop in the solutions immediately). Label all beakers that are using/will use