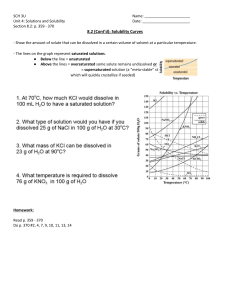
Solubility of Solutes and Crystallisation Aim: To calculate the solubility of different solutes in water and observe the formation of crystals from saturated solutions. Safety: Wear goggles to avoid solutions splashing in eyes and wear silicone gloves to protect hands from hot equipment Method: Determining solubility 1. Label a clean beaker with your group name and the solute you are testing using masking tape. 2. Weigh the empty beaker on an electronic balance. 3. Using a measuring cylinder, carefully pour 100 mL of tap water the beaker and reweigh beaker. 4. Add a spoon of a solute, quickly stir, and see if it dissolves. If it dissolves, add another spoon. Continue until the solute can no longer fully dissolve the water – this is called a “saturated solution”. 5. Reweigh the beaker and determine the mass of solute dissolved at room temperature by subtraction. 6. Place the beaker on the hot plate and heat to 100°C 7. Repeat steps 4 - 5 for hot water. Crystallisation 8. Add at least one extra spoon of undissolved solute. 9. Put a couple drops of your desired food colouring into the jar and stir. 10. If time, repeat steps 1 – 8 for a second solute. Leave one beaker with tap water only for comparison. 11. Tie a length of string to a paddle pop stick. Place the string into the jar, poke the string to get it wet and fall in place. Rest the stick on the opening of the jar. Repeat for all the jars. 12. Place jars on a windowsill or stable flat surface that won’t be disturbed. 13. Be patient and record observations. Results: a) Calculate the solubility of each solute in grams/100mL water. b) Take note of the size and shape of the crystals produced by different solutes. Discussion: • What and why are controls used in experiments? •Were all your solutes equally soluble? How could this affect how we store and use solutions? • Did all the crystals look the same? Take the same time to grow? What could make them grow faster?