Aspirin-Titration-HASPI-Student-Lab
advertisement
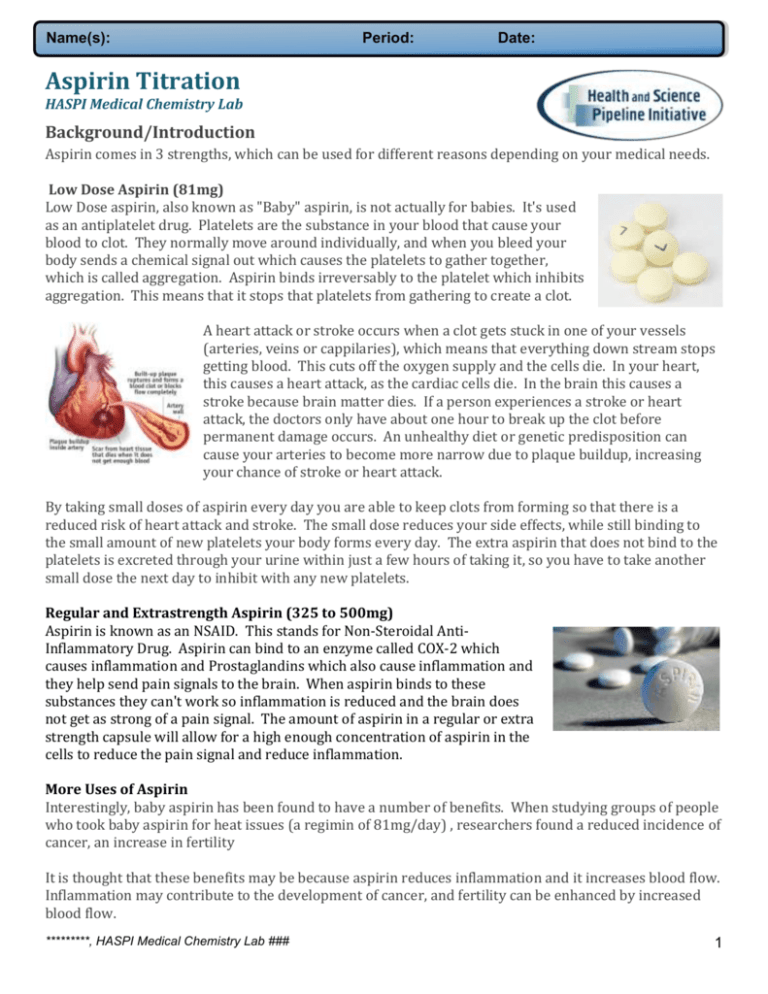
Name(s): Period: Date: Aspirin Titration HASPI Medical Chemistry Lab Background/Introduction Aspirin comes in 3 strengths, which can be used for different reasons depending on your medical needs. Low Dose Aspirin (81mg) Low Dose aspirin, also known as "Baby" aspirin, is not actually for babies. It's used as an antiplatelet drug. Platelets are the substance in your blood that cause your blood to clot. They normally move around individually, and when you bleed your body sends a chemical signal out which causes the platelets to gather together, which is called aggregation. Aspirin binds irreversably to the platelet which inhibits aggregation. This means that it stops that platelets from gathering to create a clot. A heart attack or stroke occurs when a clot gets stuck in one of your vessels (arteries, veins or cappilaries), which means that everything down stream stops getting blood. This cuts off the oxygen supply and the cells die. In your heart, this causes a heart attack, as the cardiac cells die. In the brain this causes a stroke because brain matter dies. If a person experiences a stroke or heart attack, the doctors only have about one hour to break up the clot before permanent damage occurs. An unhealthy diet or genetic predisposition can cause your arteries to become more narrow due to plaque buildup, increasing your chance of stroke or heart attack. By taking small doses of aspirin every day you are able to keep clots from forming so that there is a reduced risk of heart attack and stroke. The small dose reduces your side effects, while still binding to the small amount of new platelets your body forms every day. The extra aspirin that does not bind to the platelets is excreted through your urine within just a few hours of taking it, so you have to take another small dose the next day to inhibit with any new platelets. Regular and Extrastrength Aspirin (325 to 500mg) Aspirin is known as an NSAID. This stands for Non-Steroidal AntiInflammatory Drug. Aspirin can bind to an enzyme called COX-2 which causes inflammation and Prostaglandins which also cause inflammation and they help send pain signals to the brain. When aspirin binds to these substances they can't work so inflammation is reduced and the brain does not get as strong of a pain signal. The amount of aspirin in a regular or extra strength capsule will allow for a high enough concentration of aspirin in the cells to reduce the pain signal and reduce inflammation. More Uses of Aspirin Interestingly, baby aspirin has been found to have a number of benefits. When studying groups of people who took baby aspirin for heat issues (a regimin of 81mg/day) , researchers found a reduced incidence of cancer, an increase in fertility It is thought that these benefits may be because aspirin reduces inflammation and it increases blood flow. Inflammation may contribute to the development of cancer, and fertility can be enhanced by increased blood flow. *********, HASPI Medical Chemistry Lab ### 1 Name(s): Period: Date: Why we use Titration Titration is a way to use a very controlled reaction to quantify the amount of reactant present. In the case of medications, we can use this to determine the amount of medicine in a tablet. Because aspirin is an acid, we can neutralize it drop by drop with a known concentration of basic solution. The color will change when the reaction is complete, so that every aspirin molecule has reacted with NaOH. We can calculate the amount of aspirin using the amount of NaOH needed. An indicator is added to the solution, which will change colors when the neutralization is complete. In this case we will use phenylthalein which turns pink when it becomes basic. Companies can use this technique for quality control. Testing random medicine samples to make sure that the right concentration is going into the tablets is essential to make sure it is safe to take the medication. Additionally, a medicine can break down over time. Although aspirin has an expiration date, it actually starts to break down right away... a little at a time. A byproduct of aspirin disintigration is vinegar, so sometimes older aspirin will smell like vinegar. This byproduct can also increase side effects, so it is always best to use fresh aspirin, and it's best not to use it after the expiration date. Scientists can take a bottle of aspirin and test the tablets over time using titration to see how quickly the medicine breaks down. This helps to determine a proper expiration date for a medication, so that they can make sure that consumers are able to use it safely. PreLab Questions 1. This reading introduced new vocabulary. Using hints from the sentence they are found in or using a dictionary, define the following terms: antiplatelet inflammation aggregate excreted irreversibly inhibit vessel cardiac stroke titration heart attack indicator platelet 2. If you have a headache, what type of aspirin should you choose? 3. What are two reasons to take low dose aspirin every day? 4. Name a use for titration 5. If we had a basic medication, what kind of solution would you need in order to titrate it. 6. What kind of chemical is used to show when the neutralization is complete. *********, HASPI Medical Chemistry Lab ### 2 Name(s): *********, HASPI Medical Chemistry Lab ### Period: Date: 3 Name(s): Period: Date: Aspirin Titration HASPI Medical Chemistry Lab Objectives 1. Calculate the number of moles of aspirin in each of the tablets 2. Calculate the number of moles of NaOH needed to titrate the aspirin 3. Calculate the volume of 0.10M NaOH required to titrate the aspirin 4. Complete titrations of each aspirin to find the experimental value 4. Compare the predicted amount NaOH with the amount used experimentally Scenario The active ingredient in aspirin is acetyl salicylic acid. Different strengths of aspirin are based on the amount of active ingredients that they contain. Titration is a way to determine how much acid is in a solution by adding just enough base, of a known concentration, to neutralize the acid. At the point of neutralization, the number of moles of acid (H+) is combined with an equal number of moles of base (OH-). You will perform a titration by dispensing a base of known volume and concentration into an acid solution of known volume but an unknown concentration. The aspirin will be titrated against a standard solution of base, 0.100 M NaOH. This base will be dispensed from a burette into a beaker containing the dissolved (in ethanol) acetyl salicylic acid and phenolphthalein indicator, which appears as a faint pink color in basic solutions In this lab you will be given three types of aspirin, baby aspirin containing 81mg of aspirin, regular strength aspirin containing 325mg of aspirin and extra strength aspirin containing 500.mg of aspirin. First you will calculate the amount of NaOH you will need in order to do the titration, then you will proceed with the titration and find your error. Materials 1 baby aspirin 1 regular strength aspirin 1 extra strength aspirin Mortar & Pestle 25mL or 50mL graduated cylinder Pipette 1 buret Ring stand and buret clamp 0.10M NaOH Phenylthalein 125mL Erlenmeyer flask DI water Ethanol Predictions We are going to begin by calculating the volume of NaOH which we expect to use. The way we predict the amount of reactants needed is through stoichiometry. First take a look at the reaction: HC9H7O4 + NaOH NaC9H7O4 + H2O *********, HASPI Medical Chemistry Lab ### 4 Name(s): Period: Date: This is considered a neutralization reaction, because an acid (aspirin) and a base (NaOH) react to form a salt and water. If you look at the reaction you can see that this is balanced already with all coefficients of 1. Your goal is to calculate the number of moles of NaOH needed to neutralize each type of acid. Then you will determine how much of the 0.10M solution will be needed. Calculate the molar mass of aspirin Work: Answer Prediction: Baby Aspirin Number of mg in one tablet Number of grams in one tablet Work given Answer 81mg Work given Answer 325mg Work given Answer 500.mg Convert the grams into moles of aspirin using the molar mass Using your mole ratio, determine the moles of NaOH required to neutralize this sample of aspirin If you have a 0.10M solution of NaOH what is the volume of NaOH you will need in L and mL? (M=moles/L) Prediction: Regular Strength Aspirin Number of mg in one tablet Number of grams in one tablet Convert the grams into moles of aspirin using the molar mass Using your mole ratio, determine the moles of NaOH required to neutralize this sample of aspirin If you have a 0.10M solution of NaOH what is the volume of NaOH you will need in L and mL? (M=moles/L) Prediction: Extra Strength Aspirin Number of mg in one tablet Number of grams in one tablet Convert the grams into moles of aspirin using the molar mass Using your mole ratio, determine the moles of NaOH required to neutralize this sample of aspirin If you have a 0.10M solution of NaOH Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 5 Name(s): Period: Date: what is the volume of NaOH you will need in L and mL? (M=moles/L) Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 6 Name(s): Period: Date: Procedure/Directions Your lab team will be giving tasks, or directions, to perform on the left. Record your questions, observations, or required response to each task on the right. Task Response 1 Clean your buret. First rinse the buret with DI water, then rinse with 0.1M NaOH. Discard the water and NaOH used for rinsing. Fill your buret with 0.100M NaOH, making sure to clear the tip of the buret. 2 Grind the children's aspirin into a fine powder using the mortar and pestle. (If you have already titrated the children's aspiring move on to the regular strength and extra-strength tablets) 3 Add 10mL of ethanol to your mortar and mix with your powdered aspirin 4 Use a pipette to transfer the ethanol-aspirin solution to your Erlenmeyer flask 5 Add 25.0mL of DI water to the flask along with 2 drops of phenolphthalein indicator. Swirl to mix. 6 Record the initial volume of NaOH in the data table. 7 Begin titrating. Add NaOH in 1mL increments while constantly swirling. When the solution takes a longer time to turn clear, add NaOH drop by drop, and swirl well between drops. STOP titrating when the color change is permanent. The lighter the pink color the closer you are to the end point. 8 Record the final volume of NaOH reached during your titration on your data table 9 Clean out the Erlenmeyer flask and rinse with DI water. Repeat steps 2-7 for regular strength and extra strength aspirin samples. 10 When all three titrations are complete, discard any remaining NaOH and rinse your buret with DI water. Leave the buret clamped upside down with the tip open to dry. *Record data *Record data Data Trial 1: Baby Aspirin Initial Volume NaOH Final Volume NaOH Volume of NaOH used Trial 2: Regular Strength Aspirin Initial Volume NaOH Final Volume NaOH Volume of NaOH used Trial 3: Extra strength Aspirin Initial Volume NaOH Final Volume NaOH Volume of NaOH used Data Analysis - Calculations Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 7 Name(s): Period: Date: Results: Baby Aspirin Number of mL required to reach endpoint Number of moles NaOH titrated. Remember it was 0.10M NaOH and M=mol/L Using your mole ratio, determine the moles of aspirin neutralized by this amount of NaOH Calculate the grams of aspirin neutralized in this trial of the experiment Calculate the mg of aspirin neutralized in this trial of the experiment. Work from experiment Answer Results: Regular Strength Aspirin Number of mL required to reach endpoint Number of moles NaOH titrated. Remember it was 0.10M NaOH and M=mol/L Using your mole ratio, determine the moles of aspirin neutralized by this amount of NaOH Calculate the grams of aspirin neutralized in this trial of the experiment Calculate the mg of aspirin neutralized in this trial of the experiment. Work from experiment Answer Results: Extra Strength Aspirin Number of mL required to reach endpoint Number of moles NaOH titrated. Remember it was 0.10M NaOH and M=mol/L Using your mole ratio, determine the moles of aspirin neutralized by this amount of NaOH Calculate the grams of aspirin effectively neutralized. Calculate the mg of aspirin neutralized in this trial of the experiment. Work from experiment Answer Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 8 Name(s): Period: Date: Conclusions 1. Compare your predicted volumes of NaOH with your found volumes of NaOH using the data table below. %error = theoretical - actual x 100 theoretical Baby Aspirin Regular Strength Aspirin Extra Strength Aspirin Predicted theoretical volume of NaOH needed Actual volume of NaOH needed Percent error (show your work) 2. Acetyl salicylic acid (HC9H7O4) is a weak acid which means that hydrogen ions (H+) partially dissociate in solution. Nevertheless, the hydrogen ions that did dissociate were completely neutralized by the hydroxide ions (OH ). Describe how this can account for the percent error above. 3. Aspirin is an acid. As an acid it gives of hydrogens when put into solution. Study the reaction below and circle the hydrogen that is donated in this reaction. This is what we call the acidic hydrogen. 4. Using the following data table, calculate the cost effectiveness of each type of aspirin: Baby Aspirin Regular Strength Extra Strength Cost per bottle $11.98 $9.47 $9.29 Pills per bottle 300. 200. 100. Cost per pill Grams Effective *from your calculations Cost per pill = cost effectiveness grams effective 5. Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 9 Name(s): Period: Date: Connections & Applications As per NGSS/CCSS incorporate extensions that allow teachers/students to choose outside activities recommended by the standards. Goal of having a minimum of 3 options for every lab activity, but more can be included. May include: Write a lab procedure for the study of aspirin over time Critical thinking question(s) requiring research Medical case study related and/or reinforcing concept Investigating a phenomena Designing an experiment Developing and/or building a model Creating a multimedia or visual display to convey a concept Conduct a research project (correctly citing text is crucial) Constructing an oral or written presentation/argument (correctly citing text is crucial) Collecting data and construction graphical representations Assess accuracy and credibility of a scientific publication Mathematical computations Resources and References List of references http://www.ijpsonline.com/article.asp?issn=0250474X;year=2010;volume=72;issue=5;spage=649;epage=651;aulast=Maheshwari http://www.bostonglobe.com/lifestyle/health-wellness/2014/02/24/aspirin-may-cut-cancer-risk-but-itsbenefits-outweigh-possible-side-effects/dghiZ7q6Da17HyOIpI1KiI/story.html http://health.howstuffworks.com/medicine/medication/aspirin2.htm http://theunnecesarean.com/blog/2010/8/25/aspirin-in-early-pregnancy-may-preventpreeclampsia.html#sthash.3Whjm8fk.dpbs Aspirin Titration HASPI Medical Chemistry Lab Draft 2/28/2014 10