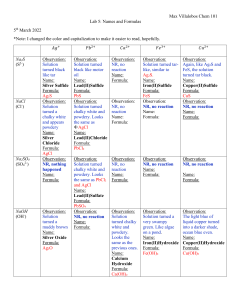
KEY Bonding and Nomenclature Test Study Guide
advertisement

Bonding and Nomenclature Study Guide Tutoring is available after school Tuesday and before school on Wednesday. You can also go to this website for assistance as well. http://misterguch.brinkster.net/ionic.html http://misterguch.brinkster.net/covalentcompounds.html http://misterguch.brinkster.net/lewisstructures.html Type III Covalent Compounds 1. oxygen difluoride OF2 2. sulfur hexafluoride SF6 3. silicon dioxide SiO2 4. SiF4 silicon tetrafluoride 5. N2O dinitrogen monoxide 6. NO2 nitrogen dioxide Type I Regular Ionic Compounds 7. sodium fluoride NaF 8. potassium sulfide K2S 9. barium cyanide Ba(CN)2 10. magnesium nitrate Mg(NO3)2 11. ammonium phosphide (NH4)3P 12. KCl potassium chloride 13. Na2O sodium oxide 14. RaClO2 radium chlorite 15. Na2CO3 sodium carbonate 16. Cs2SO4 cesium sulfate Type II Transition Metal Ionic Compounds 17. copper (I) oxide Cu2O 18. copper (II) oxide CuO 19. iron (III) sulfite Fe2(SO3)3 20. lead (IV) hydroxide Pb(OH)4 21. tungsten (VI) phosphate W(PO4)2 22. Cu2S copper (I) sulfide 23. FeO iron (II) oxide 24. MoF2 molybdenum (II) fluoride 25. Fe2(C2O4)3 iron (III) oxalate 26. PbS lead (II) sulfide Acids 27. phosphoric acid H3PO4 28. carbonic acid H2CO3 29. hydrosulfuric acid H2S 30. hydroiodic acid HI 31. chlorous acid HClO2 32. H2C2O4 oxalic acid 33. HClO hypochlorous acid 34. H2SO4 sulfuric acid 35. HBr hydrobromic acid 36. H3P hydrophosphoric acid Hodgepodge: These can by any type. 37. carbon dioxide CO2 38. potassium cyanide KCN 39. nitrous acid HNO2 40. copper (II) phosphate Cu3(PO4)2 41. disilicon trioxide Si2O3 42. hydrochloric acid HCl 43. aluminum iodide AlI3 44. BrF3 bromine trifluoride 45. Li2CO3 lithium carbonate 46. Fe3(PO3)2 iron (II) phosphite 47. HNO3 nitric acid 48. CaF2 calcium fluoride 49. HF hydrofluoric acid 50. Cu2CO3 copper (I) carbonate Ionic Bonding: Lewis Structures (Show transfer of electrons and charges formed.) 51. Li and S 52. Mg and P Covalent Bonding: Lewis Structures (Show the sharing of electrons.) 53. H2O 54. SiF4 55. SO3 56. Review from previous tests. a. Write electron configurations for: i. Fe 1s22s22p63s23p64s23d6 or [Ar] 4s23d6 ii. Si 1s22s22p63s23p2 or [Ne] 3s23p2 iii. Rb 1s22s22p63s23p64s23d104p65s1 or [ Kr]5s1 b. How many protons, electrons, and neutrons are in i. 9140Zr #p+ = 40 #e- = 40 #n0 = 91-40 = 51 ii. 227 Ac 89 #p+ = 89 #e- = 89 #n0 = 227-89 = 138