Slides - Projects In Knowledge
advertisement

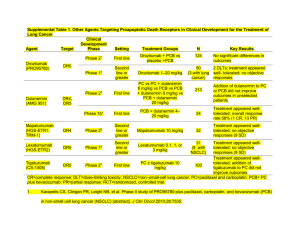
Today’s Challenges and Controversies in Recurrent Ovarian Cancer Management Bradley J. Monk, MD, FACS, FACOG Division of Gynecologic Oncology Department of Obstetrics and Gynecology Chao Family Comprehensive Cancer Center University of California, Irvine Medical Center Orange, California What is Ovarian Cancer? 3 types of cancer Epithelial Germ cell Stromal Epithelial tumors are of mesodermal origin Same as primary peritoneal cancer Epithelial cancers related to ovulatory events, which increase mutation frequency Reduced by OCPs, pregnancy, or lactation Newly Diagnosed Advanced Ovarian Cancer Courtesy of Robert Bristow, Johns Hopkins. Ovarian Cancer is a Global Disease International Agency for Research on Cancer (WHO). Courtesy of Dr. Bradley J. Monk. Ovarian Carcinoma Incidence and Mortality Incidence in US women 21,550 cases in 2009 9th most common cancer 2nd most common gynecologic cancer 1.5% lifetime risk of getting ovarian cancer Mortality in US women 14,600 deaths in 2009 5th most common cause of cancer death Most common cause of death due to gynecologic cancer 1.0% lifetime risk of dying of ovarian cancer American Cancer Society. Available at: http://www.cancer.org/ Cancer in the United States With permission from American Cancer Society. Available at: http://www.cancer.org/ FIGO Stage—Outcomes Stage Description Incidence, % Survival, % I Confined to ovaries 20 73 II Confined to pelvis 5 45 III Spread IP or nodes 58 21 IV Distant metastases 17 <5 Abbreviation: IP, Intraperitoneal Gynecologic Oncology Group database (J. Tate Thigpen). Courtesy of Dr. Bradley J. Monk. Results of Treatment Advanced Disease Parameter Small Volume Large Volume Response, % 95 75 Clinical CR, % 95 50 Pathologic CR, % 50 25 PFS, mo 25 18 Survival, mo 50 36 30–40 15–20 10-y survival, % Gynecologic Oncology Group database (J. Tate Thigpen). Courtesy of Dr. Bradley J. Monk. Results of Treatment Advanced Disease Parameter, % Small Volume Large Volume 1980: 10-y survival 7 0 1990: 10-y survival 20 10 2008: 10-y survival 30–40 15–20 Gynecologic Oncology Group database (J. Tate Thigpen). Courtesy of Dr. Bradley J. Monk. Ovarian Cancer Increasing Survival Rates Proportion Surviving SEER Data 1973-1997 N=32,845 0.75 0.70 0.65 0.60 0.55 0.50 0.45 0.40 0.35 0.30 0.25 0.20 0.15 0.10 0.05 0.00 1973-1979 1980-1989 1990-1997 2-Year Survival 5-Year Survival Barnholtz-Sloan JS, et al. Am J Obstet Gynecol. 2003;189(4):1120-1127. Approved Drugs in Ovarian Cancer 1978 2009 1978 Cisplatin 1989 Carboplatin 1990 Altretamine 1992 Paclitaxel 1996 Topotecan 1999 Liposomal doxorubicin (accelerated) 2005 Liposomal doxorubicin (full) 2006 Gemcitabine (with carboplatin) 2009 Trabectedin (with liposomal doxorubicin EMEA only) First-line Therapy Global Standard Treatment Surgery with maximum cytoreduction effort IV Platinum + Taxane Chemotherapy (Carboplatin + Paclitaxel) x 6 2004 Consensus Statements on the Management of Ovarian Cancer: Final Document of the 3rd International GCIG Ovarian Cancer Consensus Conference (GCIG OCCC 2004). Ann Oncol. 2005;16(suppl 8) viii7–viii12. Courtesy of Dr. Bradley J. Monk. Maximal Primary Cytoreduction Meta-analysis: 53 studies (1989–1998) 81 cohorts (stage III/IV) N = 6885 patients Results Expert centers have high optimal rates Optimal vs not: 11 mo (50% increase) Each 10% in cytoreduction = 5.5% in survival Platinum intensity = NS Bristow RE, et al. J Clin Oncol. 2002;20:1248-1259. Basis for Current Standard Systemic Therapy Studies showing paclitaxel/cisplatin superior to cyclophosphamide/cisplatin GOG Protocol 1111 EORTC-NCIC OV 102 Studies showing paclitaxel/carboplatin at least equivalent to paclitaxel/cisplatin in efficacy AGO Trial3 GOG Protocol 1584 1. McGuire WP, et al. N Engl J Med .1996;334:1-6. 2. Piccart MJ, et al. J Natl Cancer Inst. 2000;92:699-708. 3. DuBois A, et al. J Natl Cancer Inst. 2003;95:1320-1329. 4. Ozols RF, et al. J Clin Oncol. 2003;21:3194-3200. Standard of Care—2010 Maximum attempt at surgical cytoreduction Chemotherapy following surgery Regimen of choice Paclitaxel 175 mg/m2/3 h IV + Carboplatin AUC 6–7.5 IV Repeat every 3 wk for 6 cycles 2004 Consensus Statements on the Management of Ovarian Cancer: Final Document of the 3rd International GCIG Ovarian Cancer Consensus Conference (GCIG OCCC 2004). Ann Oncol. 2005;16(suppl 8) viii7–viii12. Courtesy of Dr. Bradley J. Monk. The Role of Antivascular Agents in the Management of Recurrent Ovarian Cancer Robert A. Burger, MD Professor, Surgical Oncology Section of Gynecologic Oncology Director, Women’s Cancer Center Fox Chase Cancer Center Philadelphia, Pennsylvania Angiogenesis in Tumor Progression With permission from The Angiogenesis Foundation, Inc; www.angio.org. Angiogenic Balance Proangiogenic Antiangiogenic VEGF FGF PGF TGFs Angiogenin Interleukin-8 HGF GCSGF PDEGF Angiopoietin 1 Thrombospondin-1 Angiostatin Interferon-alpha Prolactin 16-kd fragment Metalloproteinase inhibitors PF-4 Genistein Placental proliferin-related protein TGF-β Endostatin Hanahan & Folkman. Cell. 1996;86:353–364. Rationale for Targeting VEGF in Treatment of EOC Human tumors VEGF expression and degree of tumor angiogenesis (microvessel density) associated with Ascites formation Malignant progression1-3 Poor prognosis4-7 Abbreviation: EOC, epithelial ovarian cancer. 1. Yoneda J, et al. J Natl Cancer Inst. 1998;90:447-454. 2. Ferrara N. J Mol Med. 1999;77:527-543. 3. Dvorak HF. J Clin Oncol. 2002;20:4368-4380. 4. Gasparini G, et al. Int J Cancer. 1996;69:205-211. 5. Hollingsworth HC, et al. Am J Pathol. 1995;147:33-41. 6. Paley PJ, et al. Cancer. 1997;80:98-106. 7. Alvarez AA, et al. Clin Cancer Res. 1999;5:587-591. Rationale for Targeting VEGF in Treatment of EOC Preclinical models of solid tumors Anti-VEGF therapy Slowing of tumor progression1,2 Resolution of malignant effusions2 Synergy with cytotoxic agents3-5 Abbreviation: EOC, epithelial ovarian cancer. 1. Byrne AT, et al. Clin Cancer Res. 2003;9:5721-5728. 2. Mesiano S et al. Am J Pathol. 1998;153:1249-1256. 3. Gorski DH, et al. Cancer Res. 1999;59:3374-3378. 4. Lee CG, et al. Cancer Res. 2000;60:5565-5570. 5. Hu L, et al. Am J Pathol. 2002;161:1917-1924. Direct Anti-VEGF Antitumor Effect? VEGFRs expressed in multiple solid tumor types: colon,1 breast,2,3 ovary4 In vitro stimulation of breast carcinoma cells with VEGF3 leads to Invasion Growth factor signaling Activation of VEGFR-1 on tumor cells by VEGF3 Invasion Activation of MAPK Cell migration 1. Fan F, et al. Oncogene. 2005;24:2647-2653. 2. Wu Y, et al. AACR 2004. Abstract 3005. 3. Price DJ, et al. Cell Growth Differ. 2001;12:129-135. 4. Chen H, et al. Gynecol Oncol. 2004;94:630-635. Classes of Anti-VEGF Agents Target Ligand (VEGF) VEGFR Pharmacology Large molecules (monoclonal antibodies, soluble receptors) Small molecule inhibitors (“ibs”) Classes of Anti-VEGF Agents Target Class Agents Mab Bevacizumab1 Soluble decoy receptor VEGF Trap2,3 Ligand VEGF-A VEGF Receptor VEGFR2 Mab Ramucirumab VEGFR + PDGFR + Raf-K TKI Sorafenib VEGFR + PDGFR TKI Sunitinib VEGFR + PDGFR TKI Motesanib VEGFR + PDGFR + FGF TKI Pazopanib VEGFR + PDGFR + FGF TKI Cediranib VEGFR + PDGFR + FGF TKI BIBF-1120 Abbreviations: FGF, fibroblast growth factor; Mab, monoclonal antibody; PDGFR, platelet-derived growth factor receptor; TKI, tyrosine kinase inhibitor; VEGFR, vascular endothelial growth factor receptor. 1. Presta G, et al. Cancer Res. 1997;57:4593-4599. 2. Holash J, et al. Proc Natl Acad Sci U S A. 2002;99:11393-11398. 3. Hu L, et al. Clin Cancer Res. 2005;11:6966-6971. Unique Toxicities of Anti-VEGF Agents Proteinuria Hypertension Mucosal hemorrhage Wound healing Arterial thromboembolism Reversible posterior leukoencephalopathy syndrome (RPLS) GI perforation or fistula Single-Agent Activity Single-Agent Anti-VEGF Therapy in EOC/PPC—Phase II Efficacy Results Trial GOG 170-D1 Cannistra et al2* Tew et al3† Agent Bevacizumab Bevacizumab VEGF Trap Enrollment, n 62 44* 162‡ 1o platinum DFI <6 mo, % 36 84 47 34/66/0/0 0/52/48/0 0/0/46/46 GOG/ECOG PS (0/1/2), % 73/27/0 59/41/0 60/33/7 RR, n (%) 13 (21) 7 (16) 13(8) 40 28 4 Previous regimens (1/2/3/4), % 6 mo PFS (%) *Trial terminated prematurely. †Trial failed to meet primary endpoint. ‡Preliminary analysis. Abbreviations: EOC, epithelial ovarian cancer; PPC, primary peritoneal cancer. 1. Burger RA, et al. J Clin Oncol. 2007;25:5165-5171. 2. Cannistra SA, et al. J Clin Oncol. 2007;25:5180-5186. 3. Tew WP, et al. Paper presented at: 43rd ASCO; June 1-5, 2007. GOG 170-D Exploratory Analysis of Prognostic Factors for PFS (Proportional Hazards) GOG performance status >0 vs 0 Hazard ratio 1.491 Wald P value 0.2466 Platinum sensitivity yes vs no Hazard ratio 0.803 Wald P value 0.4702 Age Hazard ratio 1.001 Wald P value 0.9076 Number of prior regimens 2 vs 1 Hazard ratio 0.616 Wald P value 0.1207 Burger RA, et al. J Clin Oncol 2007; 25: 5165-5171. Single-Agent Anti-VEGF Therapy in EOC/PPC Toxicities—Phase II Trials GOG 170-D1 Cannistra et al2* Tew et al3† Enrollment, n 62 44* 162‡ ≥ G3 GI perforation 0 5 (11%) 2 (1.2%) ≥ G3 arterial TE 0 3§ (7%) 1 ≥ G3 HTN 6 (10%) 4§ (9.1%) 18% ≥ G3 CNS 0 1§ 1 ≥ G3 proteinuria 1 0 7% Trial *Trial terminated prematurely. †Trial failed to meet primary endpoint. ‡Preliminary analysis. §Event fatal in 1 case. Abbreviations: EOC, epithelial ovarian cancer; PPC, primary peritoneal cancer. 1. Burger RA, et al. J Clin Oncol. 2007;25:5165-5171. 2. Cannistra SA, et al. J Clin Oncol. 2007;25:5180-5186. 3. Tew WP, et al. J Clin Oncol.2007;25 (suppl). Abstract 5508. Rationale for Combination AntiVEGF and Cytotoxic Regimens Complementary, independent activity Synergy in preclinical models1 Hypothetical mechanisms Sensitization to apoptosis Reversal of cytotoxic drug resistance Increased access of chemotherapeutic—vascular “normalization”2,3 Positive phase III trials (bevacizumab) in metastatic colorectal,4,5 breast,6 and lung7 cancers 1. Sweeney CJ, et al. Cancer Res. 2001;61:3369-3372. 2. Jain RK. Science. 2005;307:5706:58-62. 3. Wildiers H, et al. Br J Cancer. 2003;88:1979-1986. 4. Hurwitz H, et al. N Engl J Med. 2004;350:23352243. 5. Giantonio B, et al. J Clin Oncol. 2007;25:1539-1544. 6. Miller K, et al. N Engl J Med. 2007:357:2666-2676. 7. Sandler A, et al. N Engl J Med. 2006;355:2542-2550. Bevacizumab + Metronomic Cyclophosphamide in Recurrent Ovarian Cancer—Phase II Results Toxicity findings similar to single agent anti-VEGF phase II trials N = 70 GI fistula/perforation: 4 (5.7%) CNS ischemia: 2 (2.9%) Wound healing impairment: 1 Pulmonary hypertension: 2 (2.9%) Treatment related deaths: 3 (4.3%) Garcia AA, et al. J Clin Oncol. 2008;26:76-82. Efficacy PFS-6 mo: 56% Response rate: 24% (17 PR) GOG 213 Recurrent ovarian and peritoneal primary cancer TFI > 6 mo Surgical candidate? Yes No Randomize Randomize Surgery No surgery Carboplatin, paclitaxel To chemotherapy randomization Abbreviation: TFI, treatment-free interval Slide courtesy of Dr. Robert A. Burger. ClinicalTrials.gov. NCT00565851 PI: Coleman, Activated 12/07 Carboplatin, paclitaxel, bevacizumab Maintenance bevacizumab Primary endpoint: Overall survival Secondary endpoints: Progression-free survival, toxicity, quality of life, translational research OCEANS—Carboplatin/Gemcitabine ± Bevacizumab in Ovarian Carcinoma (Phase III) Patients with platinumsensitive recurrent epithelial ovarian, peritoneal, or fallopian tube carcinoma (Planned N = 450) Bevacizumab + Carboplatin + Gemcitabine Placebo + Carboplatin + Gemcitabine Primary outcome measure: Progression-free survival Secondary outcome measures: objective response, duration of response, overall survival, incidence of GI perforation, safety of bevacizumab + carboplatin/gemcitabine, all adverse events Slide courtesy of Dr. Robert A. Burger. ClinicalTrials.gov. NCT00434642 Phase III Frontline Ovarian Cancer Trials Trial Sample size Arms GOG 2181 ICON-72 1800 1520 CT CT CT + bev CT + bev bev CT + bev bev Primary endpoint PFS (10/08) PFS Placebo-controlled Yes No FIGO stage III/IV High risk I/IIA, IIB-IV Secondary endpoints PFS, TOX, RR, QOL, TR OS, TOX, RR, QOL, PH-EC Bev dose/schedule 15 mg/kg every 21 days 7.5 mg/kg every 21 days Closed 7/09 Activated 4/06 Status 1. ClincialTrials.gov. NCT00262847. 2. ClincialTrials.gov. NCT00483782. Abbreviations: PFS,progression-free survival; PH-EC, pharmacoeconomics; QOL, quality of life; RR, response rate; TOX, toxicity; TR, translational research. GOG 0218—Frontline CT ± Bev in Recurrent Ovarian Cancer (Phase III) Phase B Phase A Arm 1 CT1 CT + plac 2-6 Plac 7-22 Arm 2 CT1 CT + bev 2-6 Plac 7-22 Arm 3 CT1 CT + bev 2-6 Bev 7-22 Cycles Months 1 1 2 3 2 4 5 3 ClincialTrials.gov. NCT00262847. 6 4 7 5 8 9 6 10 11 12 13 14 15 16 17 18 19 20 21 22 7 8 9 10 11 12 13 14 15 Slide courtesy of Dr. Robert A. Burger. Multi-Growth Factor Targeting The Role of the Platelet-Derived Growth Factor (PDGF) Pathway PDGF/PDGFR recruitment of pericytes1 PDGFR activation direct effect on endothelial cells2-5 Implicated in resistance to VEGF pathway inhibition6-9 1. Jain & Booth. J Clin Invest. 2003. 2. Beitz, et al. Proc Natl Acad Sci USA. 1991. 3. Risau, et al. Growth Factors. 1992. 4. Oikawa, et al. Biol Pharm Bull. 1994 5. Apte, et al. Clin Cancer Res. 2004. 6. Abramsson, et al. J Clin Invest. 2003; 7. Benjamin. Development. 1998. 8. Bergers, et al. J Clin Invest. 2003. 9. Lu, et al. Am J Obstet Gynecol. 2008. The Role of FGF in Angiogenesis FGF HGF expression (VSMC) VEGF expression (myocytes, stromal cells, EC) EC activation PDGFR expression (EC, VSMC) Mural cell recruitment Angiogenesis Abbreviations: EC, endothelial cell; FGF, fibroblast growth factor; HGF, hepatocyte growth factor; PDGFR, platelet-derived growth factor receptor; VSMC, vascular smooth muscle cell. Slide courtesy of of Dr Robert A. Burger. Murakami and Simons. Curr Opin Hematol. 2008;15:215–220. Toxicities Associated with Dual Multi-Pathway Inhibitors Dermatologic toxicity (ie, hand-foot skin reaction, rash/desquamation, skin discoloration) GI toxicity (ie, diarrhea, nausea, abdominal pain, vomiting, dyspepsia, constipation, perforation) Hypertension Fatigue Weight loss Alopecia Anorexia Asthenia Mucositis/stomatitis Altered taste Bleeding events Arterial thrombotic events Myocardial toxicity Hypothyroidism Adrenal toxicity Phase II Efficacy—MultiTargeted TKI Trial GOG 170-F Agent Sorafenib PI Matei1 N 59 GOG 170-L Motesanib Schilder 23* VEG104450 Pazopanib Friedlander2 36 PMH-PHL Cediranib Hirte3 62 4.8% ? DFCI-05170 Cediranib Matulonis4 46 17% ~ 38% BIBF- 1120 Ledermann5 83** EUDRACT RR 3.4% 6-Mo PFS 24% Other CA125 RR 31% PFS HR .68 * Closed due to toxicity ** Secondary Consolidation, Placebo-Controlled 1. Matei D, et al. J Clin Oncol. 2008;26(suppl). Abstr 5537. 2. Friedlander M, et al. J Clin Oncol. 2007; 25(suppl). Abstr 5561. 3. Hirte HW, et al. J Clin Oncol. 2008;26(suppl). Abstr 5521. 4. Matulonis UA, et al. J Clin Oncol 2009; 27: 5601-5606. 5. Ledermann JA, et al. J Clin Oncol. 2009;27(suppl). Abstr 5501. ICON 6 (Second-Line European Trial) 2:3:3 Randomization Platinum-based chemo (± taxane) q 21 days x 6 cycles + placebo Patients with platinumsensitive ovarian cancer Relapsed >6 mo following first-line platinum-based treatment Measurable disease Platinum-based chemo (± taxane) q 21 days x 6 cycles + oral cediranib daily during chemo, then 18 mo placebo Platinum-based chemo (± taxane) q 21 days x 6 cycles + oral cediranib during chemo and until progression or 18 mo Slide courtesy of Dr. Robert A. Burger. Planned: phase II (N = 300), phase III (N = 2,000). Theoretical Pros and Cons of Multi-Targeting vs Isolating VEGF Pros May more effectively block angiogenesis Potentially reduced likelihood of resistance due to activity of compensatory pathways Benefit of oral vs IV route Cons Benefit of IV vs oral Increased risk of off-target effects Vascular Disrupting Agents Unlike anti-angiogenesis agents, which inhibit the formation of new blood vessels, vascular disrupting agents destroy the existing vascular structure in a tumor Small molecule flavonoids: DMXAA Microtubulin-destabilizing agents: combretastatin (CA4P, AVE8062) Lippert JW III. Bioorg Med Chem. 2007;15: 605-615. Cai SX. Recent Pat Anticancer Drug Discov.2007;2:79101. Delmonte A, Sessa C. Expert Opin Investig Drugs. 2009;18:1541-1548. Summary Rationale for antiangiogenic ovarian cancer therapy Multiple pathways and targets Anti-VEGF drugs Single-agent activity Unique toxicity profile Multi-targeted inhibitors Single-agent activity Expanded toxicity profile Role of vascular disrupting agents Many unanswered questions The Role of Other Nonvascular Targeted Therapies in the Management of Recurrent Ovarian Cancer Bradley J. Monk, MD, FACS, FACOG Division of Gynecologic Oncology Department of Obstetrics and Gynecology Chao Family Comprehensive Cancer Center University of California, Irvine Medical Center Orange, California Active Nonvascular Targeted Agents in Ovarian Cancer Poly (ADP-ribose) polymerase (PARP) inhibitors Olaparib Antifolates Pemetrexed Farletuzumab (MORAb-003) Mammalian target of rapamycin (mTOR) inhibitors Everolimus Human epidermal growth factor receptor 2 (HER2) inhibitors Trastuzumab Pertuzumab PARP Inhibitors Targeting DNA Repair in Oncology—Rationale DNA damage frequently occurs in all cells Why is DNA repair a good target? DNA repair defects lead to increased cancer susceptibility and increased sensitivity to DNA-damaging agents Normal cells have multiple DNA repair pathways but some are lost in cancer cells Inhibiting DNA repair in cancer cells that have impaired repair pathways leads to selective cell killing and an increased therapeutic ratio Novel targeted therapeutic approach Kennedy RD, D’Andrea AD. J Clin Oncol. 2006;24:3799-3808. Courtesy of Dr. Bradley J. Monk. Types of DNA Damage and Repair Type of damage Repair pathway Bulky adducts Singlestrand breaks Doublestrand breaks Base Recombinational repair excision repair HR Repair enzymes O6alkylguanine Insertions and deletions Nucleotideexcision Mismatch Direct reversal repair repair NHEJ PARP ATM DNA-PK XP, MSH2, poly-merases MLH1 AGT Hoeijmakers JHJ. Nature.2001;411:366-374. Khanna KK, Jackson SP Nat Genet. 2001;27:247-254. Sanchez-Perez I. Clbl. Transl Oncol. 2006;8:642-646. Courtesy of Dr. Bradley J. Monk Targeted Killing of Cancer Cells with Defective DNA-Repair Mechanisms Double-stranded break Cancer cell with defective repair Normal cell Repair by HR pathway BRCA deficient or deficiency of other HR proteins No repair (no HR pathway) Survival Cell death Exploits inherent weakness of cancer cells that have defective DNA repair Abbreviation: HR, homologous recombination. Martin SA, et al. Curr Opin Genet Develop.2008;18:80-86. Courtesy of Dr. Bradley J. Monk. DNA Repair Inhibitors in Cancer Cells Two Modes of Action Potentiation Inhibition of DNA repair following DNA-damaging agents Original hypothesis Synthetic lethality Selected cancer cells lose DNA repair pathways, whereas normal cells remain unaffected Targeting these defective cells may cause selective cell kill with an increased therapeutic ratio May allow for a novel targeted approach to cancer treatment Bentle MS, et al. J Mol Histol. 2006;37:203-218. Olaparib (AZD 2281) Development Oral small molecule PARP inhibitor (low nM) Escalation phase (N = 46) All tumors BRCA mutation not required (11 BRCA ovarian cancers) 10 dose levels; administration 2 of 3 weeks up to BID continuously PK and PD determined DLT: Myelosuppression, N/V, CNS (mood changes) MTD: 400 mg BID Expansion phase (N = 52) All confirmed BRCA mutation carriers (39 ovarian cancers) DLT: Fatigue, thrombocytopenia, somnolence Administration 200 mg BID continuously Yap TA, et al. J Clin Oncol. 2007;25(suppl). Abstr 3529. Fong PC, et al. J Clin Oncol. 2008;26(suppl). Abstr 5510. Phase I—Olaparib (AZD 2281) Updated Expanded BRCA Cohort Characteristic Mutation – BRCA1 – BRCA2 – Family history Age mean (range) PS (0–1) Time from Dx to Rx Platinum status – Sensitive – Resistant – Refractory Number of priors (range) Number 15 1 1 54.8 years (19–82) 55 4.7 years (0.5–16) 10 (20%) 27 (54%) 13 (26%) 3 (1–8) Fong PC, et al. N Engl J Med. 2009;361:123-134. Fong PC, et al. J Clin Oncol.2008;26(suppl). Abstr 5510. Courtesy of Dr. Bradley J. Monk. Phase I—Olaparib (AZD 2281) Clinical Activity—RECIST + GCIG Total Platinum Sensitive Platinum Resistant Platinum Refractory 46 10 25 11 Responders by RECIST 13 (28%) 5 (50%) 8 (32%) 0 (0%) Responders by GCIG CA125 18 (39%) 8 (80%) 8 (32%) 2 (18%) Responders by either RECIST or GCIG criteria 21 (46%) 8 (80%) 11 (44%) 2 (18%) SD (>4 cycles) 6 (13%) 1 (10%) 4 (16%) 1 (95) Median duration of response in weeks (range) 24 (10– 77) 23 (16–77) 24 (10–65) 26 (20–32) No. of evaluable patients Abbreviations: GCIG, Gynecologic Intergroup ;RECIST, Response Evaluation Criteria in Solid Tumors. Fong PC, et al. N Engl J Med. 2009;361:123-134. Fong PC, et al. J Clin Oncol.2008;26 (Suppl). Abstr 5510. Courtesy of Dr. Bradley J. Monk. Phase II Trial of the PARP Inhibitor Olaparib (AZD 2281) in BRCA-Deficient Advanced Ovarian Cancer—Efficacy Patients with confirmed mutation, recurrent (stage IIIB/IIIC/IV) ovarian cancer after failure of ≥1 platinum-based chemotherapy Olaparib 400 mg BID (n = 33) Olaparib 100 mg BID (n = 24) Response by RECIST 11 (33%) 3 (13%) Platinum sensitive 1/7 (14%) 2/8 (25%) Platinum resistant 10/26 (38%) 1/16 (6%) 20 (61%) 4 (17%) 290 days (126–513) 269 days (169–288) 5.8 months 1.9 months (n = 33) (n = 24) Nausea 2 (6%) 3 (13%) Vomiting 2 (6%) 2 (8%) Discontinuation due to AEs 4 (12%) 1 (4%) Dose interruption due to AEs 12 (36%) 4 (17%) Response by RECIST and/or GCIG Median DOR (range) Median PFS Grade 3/4 AEs Audeh MW, et al. Presented at 45th Annual ASCO; May 29-June 2, 2009. All Patients—Who Will Benefit From PARP Treatment? Sporadic tumors with intact BRCA function Abbreviation: Nl, Normal lymphocytes. Prevention? Courtesy of Dr. Robert Coleman. Coleman RL. Curr Oncol Rep. 2009;11:414-416. Olaparib (AZD 2281) Maintenance Trial Histologically or cytologically confirmed serous epithelial ovarian cancer CR/PR to 2nd- or 3rd-line platinum-based chemotherapy (penultimate treatment-free interval >6 months) BRCA mutation not required R A N D O M I Z E Olaparib PO 400 mg BID until disease progression Placebo until disease progression Primary end point = PFS N = 250 Recruitment complete (results expected late 2010) ClinicalTrials.gov Identifier: NCT00753545. Courtesy of Bradley J. Monk. Key Issue for Future Developments of PARP Inhibitors Is single-agent PARP inhibitor or combination preferable? Single-agent treatment utilizes selective synthetic lethality with limited toxicity Combination with DNA-damaging agents (temozolamide or platinum) reverses resistance in experimental models More myelotoxicity (BSI-201 exception) What determines resistance to PARP? Return of BRCA function through intragenic deletion1 Will PARP exposure impact response to further chemotherapy? 1. Edwards SL, et al. Nature. 2008;451:1111-1115. PARP Inhibitors—Summary Active in those with BRCA germ line mutations Potential activity in those with BRCA dysfunction Synthetic lethality represents new paradigm in therapeutic oncology Combinations of PARP inhibitors and chemotherapy ongoing Antifolates Pemetrexed O O N H OH Approved in malignant mesothelioma and nonsquamous NSCLC1 Enters via reduced folate carrier and a selective high capacity transporter Active against DHFR, TS, GARFT O O OH N N H GOG 126-Q 21% RR2 HN H2N Antifolate Combination3 N-[4-[2-(2-amino-3,4-dihydro-4-oxo-7Hpyrrolo[2,3-d]pyrimidin-5yl)ethyl]benzoyl]-L-glutamic acid Pemetrexed 1. Alimta® PI, Eli Lilly and Company Indianapolis, IN, 2009. 2. Miller DS, et al. J Clin Oncol. 2009;27:2686-2691. 3. Horowitz NS, et al. J Clin Oncol. 2008;26(suppl). Abstr 5523. Abbreviations: DHFR, dihydrofolate reductase; GARFT, glycinamide formyltransferase; TS, thymidylate synthase. Targeting the Folate Receptor— Farletuzumab (MORAb-003) Humanzied MoAb to folate receptor-a (FR-a) Induces complement-dependent cytotoxicity (CDC) and antibody-dependent cell-mediated cytotoxicity (ADCC) Blocks Lyn kinase-(P) Farletuzumab (MORAb-003)—Phase II EOC in first relapse platinum-sensitive (>6 mo) Increased CA-125; Measurable or CA-125 No symptoms of disease Arm A Single-agent farletuzumab Until progression or symptoms Increased CA-125; Measurable or CA-125 Needing chemotherapy Symptoms Arm B Front-line CT regimen Farletuzumab (6 cycles) Arm C Maintenance Farletuzumab Armstrong DK, et al. Presented at 44th Annual ASCO; May 30-June 3, 2008. Courtesy of Dr. Bradley J. Monk. Farletuzumab (MORAb-003)— Phase II Response: CA-125 criteria 41 evaluable 37 (90%) normalized CA-125 34 still on study 12 on study longer than TFI1 – 6 (50%) have TFI2 > TFI1 22 in follow-up – 15 in remission – 7 relapsed Response: RECIST Independent review (early) CR: 7% PR: 63% SD: 26% PD: 4% RR: 70% Abbreviations: TFI1, first tumor-free interval; TFI2, second tumor-free interval. Armstrong DK, et al. Presented at 44th Annual ASCO; May 30-June 3, 2008. Farletuzumab (MORAb-003)— Phase II Safety Infrequent infusion reactions No additive toxicity with carboplatin/taxane Grade 3 (single agent): Headache Grade 3 (combination): Neutropenia, diarrhea Armstrong DK, et al. Presented at 44th Annul ASCO; May 30-June 3, 2008. Farletuzumab (MORAb-003)— Phase III Histologically or cytologically confirmed nonmucinous epithelial ovarian cancer including primary peritoneal or fallopian tube malignancies Measurable disease by CT or MRI scan Relapse within ≥6 and <24 mo after first-line platinum/taxane chemotherapy Candidate for repeat carboplatin/taxane therapy Neurologic function: Neuropathy (sensory and motor) ≤CTCAE Grade 1 R A N D O M I Z E Carboplatin and taxane with placebo Carboplatin and taxane with MORAb-003 1.25 mg/kg Carboplatin and taxane with MORAb-003 2.5 mg/kg Primary end point = PFS N = 900 Abbreviation: CTCAE, common terminology criteria for adverse events. US NIH. 2009. Courtesy of Dr. Bradley J. Monk. mTOR Inhibitors The PI3K/Akt/mTOR Pathway Growth factors and other mitogens PI3K Ras Akt Raf mTOR MEK Raptor Rheb GTP Nutrients, ATP AMPK Rheb GDP Phosphatases ERK 4E-BP1 Mnk-1 TSC1 TSC2 Rictor p38 LKB1 PTEN S6K pdcd4 eIF4E S6 eIF4B Translation, ribosome biogenesis, metabolism, cell growth, angiogenesis, ↓ autophagy Meric-Bernstam F, Gonzales-Angulo AM. J Clin Oncol. 2009;27:2278-2287. Courtesy of Dr. Bradley J. Monk. GOG Future Directions Randomized Phase II Ovarian Cancer— GOG 186G mTOR Inhibitor, Everolimus Regimen I: Ovarian, fallopian tube or primary peritoneal cancer 1-3 priors Bevacizumab 10 mg/kg IV q2 wks Plus Everolimus 10 mg orally daily Regimen II: Bevacizumab 10 mg/kg IV q2 wks Plus Matched placebo *Primary endpoint PFS ClinicalTrials.gov NCT00886691. Courtesy of Dr. Bradley J. Monk. HER2 Inhibitors Trastuzumab and Pertuzumab Bind Distinct Epitopes on HER2 Trastuzumab requires HER2 overexpression for activity; pertuzumab does not require overexpression Pertuzumab specifically binds HER2’s dimerization domain, which inhibits downstream signaling Trastuzumab in Ovarian Cancer HER2 expression occurs at low levels in advanced, recurrent ovarian cancer (6.7%–11.7%)1,2 Single-agent trastuzumab resulted in response rate of only 7.3% in HER2+ patients1 Trastuzumab in combination with paclitaxel and carboplatin was tested in 321 patients with advanced ovarian cancer2,3 Only 22 were HER2+, only 7 met the eligibility criteria of the trial 3/7 had complete responses lasting 6,7+, and 24+ months 2/7 had stable disease 1. Bookman MA, et al. J Clin Oncol. 2003;21:283-290; 2. Guastalla JP, et al. J Clin Oncol. 2007;25 (suppl). Abstr 5559. 3. Ray-Coquard I, et al. Clin Ovarian Cancer. 2008:1:54-59.. Gemcitabine ± Pertuzumab Study Schema for TOC3258g Platinum-Resistant Ovarian Cancer, N=130 1:1 randomization Gemcitabine + Placeboa, n = 65 Disease Progression Discontinue Study Treatment, Cross-Over Allowed aUp to 17 cycles Makhija S, et al. J Clin Oncol. 2010;28;1215-1223. Gemcitabine + Pertuzumaba, n = 65 Disease Progression Discontinue Study Treatment HER3 mRNA Biomarker Analysis mRNA expression analysis of archival formalin-fixed paraffin-embedded tissue (FFPET) samples that focused on selected HER receptors and their ligands (EGFR, HER2, HER3, amphiregulin, and betacellulin) was performed on 122/130 (94%) of patients’ tumors; the results were correlated with clinical outcomes For these 5 markers, only HER3 expression levels correlated with differential treatment benefit between gemcitabine + pertuzumab and gemcitabine + placebo Makhija S, et al. J Clin Oncol. 2010;28;1215-1223. Gemcitabine + Pertuzumab Response Rate Gem Gem + Pertuzumab 3/65 (4.6%) 9/65 (13.8%) 2.4% 16.7% Low HER3 (<median) 0 6 High HER3 (≥median) 3 3 All patients 1 prior platinum-based Tx only (n = 90) Makhija S, et al. J Clin Oncol. 2010;28:1215-1223. Progression-Free Survival (PFS) by HER3 Median (Mo) a Gem Gem + Pertub HR P Value All patients 2.6 2.9 0.66 .07 Low HER3a 1.4 6.6 0.16 .0002 High HER3b 5.5 2.8 1.68 .0844 <50th percentile percentile b >50th Makhija S, et al. J Clin Oncol. 2010;28:1215-1223. Overall Survival by HER3 Median (Mo) a b Gem Gem + Pertub HR P Value All patients 13.1 13.0 0.91 .65 Low HER3a 8.4 12.5 0.61 .1026 High HER3b 18.2 15.1 1.59 .1943 <50th percentile >50th percentile Makhija S, et al. J Clin Oncol. 2010;28:1215-1223. Synopsis Experimental data demonstrate that HER3-HER2 signaling leads to the downregulation of HER3 mRNA in model systems The impetus to study this attenuation mechanism was driven by the analysis of HER3 mRNA levels in the TOC3258g trial This mechanism may explain pertuzumab’s activity in Pt-resistant ovarian cancer patients whose tumors express low levels of HER3 mRNA Key Takeaways Agents other than antiangiogenesis molecules active in ovarian cancer Understanding tumor biology key Rationale clinical design critical Major challenges remain Patient selection Strategic combinations Faster and cheaper development Off-Target Effects of “Targeted” Therapy Robert L. Coleman, MD Professor and Director, Clinical Research Department of Gynecologic Oncology M. D. Anderson Cancer Center Houston, Texas Targeted Agents Monoclonal Abs Tyrosine Kinase Inhibitors Long t1/2 Short t1/2 Targeted Promiscuous IV (SQ) PO (IV) Few drug-drug interactions Many drug-drug interactions Toxicity to On- and Off-Target Constituents On-Target AE’s Hypertension CNS Proteinuria GI toxicity Thyroid Perforation, fistula Hemorrhage Cardiac Off-Target AE’s Endocrine Low: Mg++, Ca++, phosphate, sodium High: glucose, alkaline phosphatase, bilirubin, transaminases Pulmonary Bleeding Congestive heart failure, conduction abnormalities Thyroid VTE Arterial and venous Abbreviation: VTE, venous thromboembolism. Electrolyte Dermatologic Rash Wound disruption Anti-Vascular Endothelial Growth Factor (VEGF)-Induced Hypertension Sorafenib: all grade hypertension ranges 16.2%–42.6% (mean: 23.4%)1 Sunitinib malate: all grade hypertension incidence 30%2 Bevacizumab: the need for antihypertensive drug therapy occurs in 10%–20% of patients (10 mg/kg) 3-5 Dose-effect: 3%–32% in low (3-7.5 mg/kg) vs 18-36% in high dose (10-15 mg/kg)6 1. Wu S, et al. Lancet Oncology. 2008;9:117-123. 2. Sunitinib PI. Pfizer, New York, NY, 2008. 3. Hurwitz H, et al. N Engl J Med. 2004; 350:2335-2342. 4. Ranpura V, et al. Am J Hypertens.2010; epub ahead of print. 5. Chowdhury S, et al. Targeted Oncology. 2006;1:104-108. 6. Gressett S, et al. Ann Pharmacother. 2009;43:490-501. Possible Mechanisms The mechanism of action by which VEGF inhibitors cause hypertension is uncertain VEGF is a stimulator of nitric oxide, and the inhibition of VEGF may cause increased systemic vascular resistance1 It has been suggested that hypertension is a biomarker for activity 1. Hood JD, et al. Am J Physiol Heart Circ Physiol. 1998;274:1054-1058. Anti-VEGF-Induced Hypertension The characteristics of hypertension caused by sorafenib was investigated in patients who sustained a systolic blood pressure elevation of ≥20 mm Hg 3 weeks after therapy initiation Pulse wave velocities and aortic augmentation indices were increased, indicating increased vascular stiffness Veronese ML, et al. J Clin Oncol. 2006;24:1363-1369. Essential Hypertension (BP >140/90) Thiazide-(like) diuretics Angiotensin converting enzyme inhibitors/ angiotensin receptor blockers Calcium channel blockers Beta blockers Essential Hypertension Each of these categories is roughly equally effective (works in 30–50% of cases) Start with single agent if blood pressure <20/10 above goal African Americans/elderly do better with diuretic/ calcium channel blocker Essential Hypertension Thiazide-like diuretics: hypokalemia, glucose intolerance, hyperuricemia, lower urinary calcium excretion Dihydropyridine calcium channel blockers: pedal edema 10% of patients on amlodipine at 10 mg develop edema1 Angiotensin converting enzyme inhibitors: cough, increased potassium levels, hyperkalemia, renal insufficiency, angioedema (↑in African-American women2) 18% of patients discontinued for toxicity,3 including 3%–11% for cough2 Beta blockers: may be associated with a small absolute increase in stroke risk 1. Amlodipine PI. Pfizer, New York, NY. January 2010. 2. Elliott WJ. Clin Pharmacol Therapeutics. 1996;60:582588. 3. Lau E. COMPETE III team. Proc CAPT, May 27-30, 2007. Essential Hypertension If first agent does not succeed, options include Increasing dose to maximum Switching agents Adding a 2nd agent Most patients with blood pressure more than 20/10 above goal will require 2 agents Essential Hypertension Useful combinations ACE inhibitors with diuretic or CCB (decreases pedal edema) Less useful CCB with diuretic Abbreviations: ACE, angiotensin converting enzyme; CCB, calcium channel blockers. Antihypertensive Therapy Non-dihydropyridine CCBs interact (inhibit) with CYP3A41, which metabolizes sorafenib2 and sunitinib3 Therefore verapamil and diltiazem (inhibitors) should be used cautiously1,4 1. 2. 3. 4. Most dihydropyridine CCBs (eg, amlodipine, nifedipine) are CYP2A4 substrates, but not inhibitors Lim GE, et al. Exp Clin Cardiol. 2003;8:99-107. Sorafenib PI. Bayer Healthcare Pharmaceuticals; Wayne, NJ. 2009. Sunitinib PI. Pfizer, Inc.; New York, NY. 2008. Bailey DG, et al. Br J Clin Pharmacol. 1998;46:101-110. Reversible Posterior Leukoencephalopathy Syndrome Courtesy of Dr. Carolyn Muller, University of New Mexico. Reversible Posterior Leukoencephalopathy Syndrome Presents with headache, seizure, lethargy, confusion, blindness Mild to severe hypertension may be present, but is not necessary for diagnosis Diagnosis is confirmed with MRI; white matter abnormalities suggestive of edema as seen in posterior parieto-occipital regions Most patients recover Bevacizumab—Clotting Arterial thrombotic events including myocardial infarction, cerebral vascular accident, arterial thromboembolic event 8.5% of patients age >65 years and 2.1% of patients age <65 years experienced arterial thromboembolic event1 1. Bevacizumab PI. Genentech, Inc., South San Francisco, CA. 2009. Bevacizumab—Bleeding Delays wound healing T1/2 ~20 days1 Minor nose bleeds common ≥5% of lung cancer patients have developed serious/fatal hemoptysis;2,3 limited data in ovarian patients 1. Bevacizumab PI. Genentech; South San Francisco, CA. 2009. 2. Johnson DH, et al. J Clin Oncol. 2004; 22:2184-2191. 3. Sandler A, et al . N Engl J Med. 2006;355:2542-2550. Fistula Management strategies • Stop agent • Conservative: Isolation (ostomy bag), NPO, TPN, octreotide (if high output) • Refractory or nonhealing: Surgery (wait 2–4 half-lives for washout) Abbreviations: NPO, nothing by mouth; TNP, total parenteral nutrition. Courtesy of Dr. Robert L. Coleman. Proteinuria Incidence of proteinuria in the bevacizumab trials for colorectal cancer has been reported as 23%–38%, compared with an incidence of 11%–22% in control groups treated with chemotherapy alone1 Incidence in ovarian cancer unknown but likely to be similar to that in colorectal cancer Development of proteinuria is associated with hypertension 1. Gordon MS, Cunningham D. Oncology. 2005;69(suppl 3):25-33. Courtesy of Dr. Robert L. Coleman. Cutaneous Toxicities of EGFR/VEGFR Inhibitors Produced by EGFR TKIs (eg, erlotinib) and MoAbs (eg, cetuximab) and VEGFR TKIs (eg, sorafenib, sunitinib, cediranib, etc) Include Acneiform rash (face, upper back) Dry itchy skin, dry mucosa Trichomegaly (long curly eyelashes) Paronychia Abbreviations: EGFR, epidermal growth factor receptor ; MoAbs, monoclonal antibodies; TKIs, tyrosine kinase inhibitors; VEGFR, vascular endothelial growth factor receptor. Courtesy of Dr. Robert L. Coleman. Cutaneous Toxicities of EGFR Inhibitors Treatment of rash Topical agents (eg, clindamycin 1% gel) Systemic antibiotics (eg, tetracycline or minocycline) Treatment of paronychia Difficult Wide shoes, good nail hygiene Local antibiotics (preventive) Systemic antibiotics (if impetiginization occurs) Sorafenib Rash Less common than and different in appearance from EGFR rashes May disappear spontaneously after several weeks of treatment Should be differentiated from classic erythrodermic allergic reactions (which may also occur with sorafenib and sunitinib) Sorafenib Rash Slide courtesy of Beth Manchen, University of Chicago. Treatment of Anti-VEGFR TKI HandFoot Syndrome Drug holiday or dose reduction Cotton socks, soft padded shoes Moisturizers Urea and/or salicylate-containing creams for calloused areas (under socks and gloves overnight) Sunitinib—Hypothyroidism 3%–4% of patients on placebo-controlled GIST trial developed hypothyroidism1 In 1 study, 85% of patients treated with sunitinib developed at least 1 laboratory abnormality c/w hypothyroidism2 Less common with sorafenib Unclear how much this contributes to fatigue 1. Sunitinib PI. Pfizer Labs. New York, NY 2010. 2. Rini BI. J Natl Cancer Inst. 2007;99:81-83. Laboratory Abnormalities 13% of patients on sorafenib developed grade 3 hypophosphatemia1 Sunitinib may cause hyperbilirubinemia2 Cetuximab/panitumumab can cause severe hypomagnesemia3,4 1. Sorafenib PI. Bayer Healthcare Pharmaceuticals Inc; Wayne, NJ. 2009. 2. Sunitinib PI. Pfizer, Inc; New York, NY 2008. 3. Cetuximab PI. ImClone Systems & Bristol-Myers Squibb; Branchburg, NJ. 2010. 4. Panitumumab PI. Amgen; Thousand Oaks, CA. 2009. QT Prolongation Most common cause of delays in drug development, nonapprovals, postmarketing withdrawals by FDA Risk of malignant cardiac arrhythmia with torsades de pointes and sudden cardiac death QT Prolongation Ion channel malfunction results in excess positive intracellular change This extends ventricular repolarization and results in a prolonged QT interval Upper limit of normal for QTc is 450 ms in men, 460 ms in women QT Prolongation—Targeted Oncology Agents Depsipeptide (HDAC inhibitor) Sunitinib (uncertain significance) Dasatinib (Src/bcr-abl/kit kinase inhibitor) Vandetanib (VEGFR/EGFR inhibitor) Torsades de Pointes http://www.aic.cuhk.edu.hk/web8/Hi%20res/torsades1.jpg Torsade de Pointes Lists of medications implicated in QT prolongation are maintained on internet by University of Arizona (www.torsades.org) CYP3A4 inhibitors may prolong t1/2 of QT prolonging drugs Grapefruit Juice Potent intestinal CYP3A4,1,2,3 CYP1A2 and CYP2A6 inhibitor Small bowel enterocyte CYP3A4 protein levels decrease 62% after 5 days of grapefruit juice2 Seville oranges have similar effect2,3 Furanocoumarins may be responsible) Absorption of drugs is therefore increased Cmax of drugs, such as rapamycin, is increased 150%–250%4 1.Yoshida M, et al. Pharmacoepidemiol Drug Safety. 2008;17:70-75. 2. Bailey DH, et al. Br. J Clin Pharmacol. 1998;46:101-110. 3. Lim GE, et al. Exp Clin Cardiol. 2003;8:99-104. 4. Cohen E, et al. Paper presented at: 100th Annual AACR; April 18-22, 2009. Adverse Events Associated with Emerging Agents in Ovarian Cancer PARP-1 inhibitor Olaparib 400 mg BID (N = 60)1 Grades 3 and 4 in >10% of patients: lymphopenia; nausea/vomiting; dizziness Folate inhibitors Pemetrexed 900 mg/m2 q21d (N = 51)2 Grades 3 and 4: neutropenia (42%); leukopenia (25%), anemia (15%); constitutional (15%) Farletuzumab 100 mg/m2 weekly (N = 28)3 Grade 3 headache as part of infusion DLT: Not reached at 400 mg/m2 the highest dose tested mTOR inhibitor Everolimus (in endometrial cancer)4 Grades 3 and 4 in >10%: fatigue; nausea; anemia; lymphopenia; electrolyte abnormalities 1. Fong PC, et al. N Engl J Med. 2009;361:123-134. 2. Miller DS, et al. J Clin Oncol. 2009;27:2686-2691. 3. Armstrong DK, et al. Presented at: 44th Annual ASCO; May 30-June 3, 2008. 4. Slomovitz BM, et al. Presented at 44th Annual ASCO; May 30-June 3, 2008. Conclusions Targeted therapies are not nontoxic Many toxicities are dose-dependent Not all toxicities are known Particularly as antiangiogenic drugs are used in earlier stage disease, careful blood pressure monitoring is essential With oral agents, drug-drug interactions are common