Thermal Physics - Issaquah Connect
advertisement

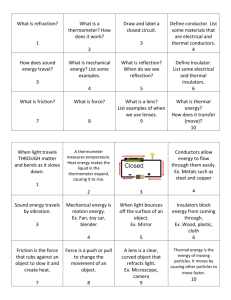
Print off topic outline by Monday. Test on this unit will be on Friday, Feb. 10 You will be completing 2-3 labs between now and then…prepare for a fast unit! Textbook Reference: Chapters 10 and 11. You WILL need your textbook every day for the next 2 weeks. Bring one or share one. When you think of Heat, Thermal Energy, and Temperature, what comes to mind? 3 min: write on your own warm-up sheet (a new one! It’s 2nd semester!) The rest of this power point is filled with guiding questions (and online resource links) to help you gather information for all the syll. Statements for this unit Take good notes (on your own…) related to each of the questions on the slides. You will NOT be getting lectures in class for this unit. There are, however, the presentations I used last year posted online—use those as supplemental notes We WILL be doing 2 labs next week. All syllabus statements must be completed by the beginning of class on Friday, Feb. 10 Please check WebAssign. There will be several relatively short homework sets, related to major topics in this unit. All will be open until 7:25 AM on Friday, Feb. 10 (the day of the test). Don’t forget to do any of them! What are the units of temperature? Which units of temperature are metric? Which is the fundamental metric unit? How do you convert between each of the temperature units? Use the following resources: Resource #1 Resource #2 Resource #3 HW#1: WebAssignThermal Physics 1: Temperature Conversions What IS absolute zero? How was it determined? What is the temperature of Absolute Zero in the Celsius temperature scale? Resource A Resource C Resource B Resource D What does “Thermometric” mean? There are several examples of these properties. List and describe each of them. Resource #4 Homework #2: (due to turnitin.com no later than Friday, Feb. 10, 2012) Design Lab: Design a lab to study the effect of temperature on a variable related to your choice of thermometric property. (please be specific about which thermometric property your are studying) Use the IB “Design” rubric Resource #5 Resource #6 Resource #8 Resource #7 If you were to construct a thermometer, why must you have 2 known, standard (fixed) points with which to work? Describe HOW you would calibrate (create the accurate scale) a standard alcohol thermometer that was originally blank (no scale on it at all) Define the following: Thermal Energy (internal energy): Heat: Temperature: Resource #9 Resource #10 Resource #11 How is the direction of thermal energy transfer determined? (3.1.1) Sketch and interpret a kinetic energy distribution for a substance at various temperatures (see last year’s notes… OR… Resource E Resource F Define/Describe each of the following: Conduction Convection Radiation Note: concepts only—you don’t need to do any calculations for any of these methods of energy transfer Resource #12 Resource #13 (also see Resource #10) What is a mole? How is molar mass of an element determined? (note: if you don’t have a periodic table, let Ms. Fowler know…) How is molecular molar mass determined? What is Avogadro’s number? Where did it come from? Resource #14 Resource #15 What is meant by “Heat Capacity”? What variables will affect the heat capacity of a substance? Mathematically, how is heat capacity determined? With what units? Resource #16 Resource #17 How is specific heat capacity “specific”? Mathematically, how is specific heat capacity determined? With what units? Resource #18 Resource #19 Resource #20 HW reminder! Calorimetry lab (data table and analysis questions A-F) due on Friday, 2/10/12! Sample Problems: there is a Word Document on both websites with practice problems (and answers)—not required to complete all of them, but 3.2.2 asks you to complete some problems…I’d recommend 2-3 be put in your syll. state. Book. HW reminder: WA Thermal problems #2— Calorimetry—is available to complete As is HW#3 (Molar mass practice) There are 3 primary phases of matter (we’re not concerned with plasmas or Bose-Einstein condensates at this point). Describe (and compare/contrast) each based on the following characteristics: (use last year’s notes) Molecular motion (i.e. kinetic energy—quantity and type) Macroscopic properties (see Word document in “notes” section online) Complete the following flow chart: Energy Added Solid Liquid Energy Removed Gas What is meant by “Latent Heat”? How is this different than “specific heat”? What happens to molecules of a substance while undergoing a change of phase? What happens to the temperature of a substance while undergoing a change of phase? Sketch a heating (or cooling) curve for water, showing the relationship between the temperature of water and the time that passes as thermal energy is being added (or removed) at a constant rate Heating curves should have proper labels showing: Melting/freezing point Boiling/condensing point State of matter or process occurring during each section of the graph What’s the difference? Which one occurs AT the boiling point? Which one occurs BELOW the boiling point? Look at last year’s notes for the required summary of the “rules” an ideal gas will follow under this model Gas Laws: Boyle’s law (this was your lab last week) ▪ Resource Resource Charles’ Law ▪ Resource Combined Gas law ▪ Resource(pHet simulation)