Water - csfcbiology
advertisement

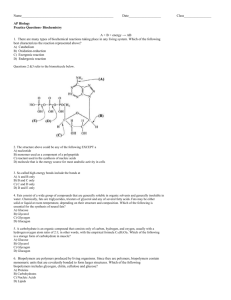
Biological Molecules: Water AS Biology: BY1 Structure of water Water (H2O) consists of two hydrogen atoms covalently bonded to one oxygen atom. Each hydrogen shares a pair of electrons with the oxygen. The oxygen has a greater affinity for electrons than the hydrogens, so it ‘pulls’ the electrons closer. δ– δ+ 104.5 ° δ+ This makes the oxygen slightly negative (indicated by δ–) and the hydrogens slightly positive (indicated by δ+). This creates different charged regions, making water a polar molecule. Because it has two charged regions it is dipolar. Hydrogen bonds Many of the properties of water are due to its ability to form hydrogen bonds. The slight negative charge on the oxygen atom makes it attract the slightly positive hydrogen atom of another water molecule. hydrogen bond The numerous hydrogen bonds in water make it a very stable structure. Inorganic Compounds: Water Polar molecule Makes a good solvent (polar substances dissociate forming solutes) form of key elements for many organisms Good for transport-not viscous Good reaction medium Adheres to surfaces (to aid transport in xylem) Figure 2.4a Water as a solvent Properties and biological roles of water Inorganic Compounds: Water Hydrogen bonding between water molecules (weak bonds) Creates cohesion (sticking to each other) Water high surface tension High Specific Heat Capacity (temperature buffer-good for organisms and enzymes) High Latent Heat of Evaporation (for cooling by sweating) Figure 2.4b Water H+ and OH participate in chemical reactions Maximum density at 4oC Allows ice to form on top thus insulating lakes/ponds Prevents all of lake from freezing and killing organisms Transparent (light can get through for photosynthesis) Multiple-choice quiz 9 of 30 © Boardworks Ltd 2008 What’s the carbohydrate? 10 of 30 © Boardworks Ltd 2008