gr12nomenclatureisu
advertisement

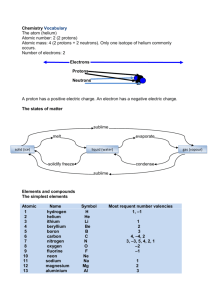
1 SCH 4U Nomenclature Independent Study An excellent knowledge of Chemical Nomenclature is essential for success in grade 12 Chemistry. For your first independent study you will review the nomenclature you learned in grade 11 and write a test. Nomenclature Test Date:___________________________________________ Valence Value The valence value of an element is defined as the numbers of electrons an atom of that element will gain/lose/share when forming a compound. Cross-Over Rule for Writing Formulas STEPS 1. Write down the symbols of the elements in the order given in the name. 2. Write the valences above the elements symbol. 3. Divide the valences by the highest common multiple. 4. Cross-over valences 5. Drop all 1’s and unnecessary brackets sodium chloride magnesium oxide calcium fluoride lithium sulphate aluminum phosphate Na Cl Mg O Ca F Li SO4 Al PO4 1 1 Na Cl 1 1 Na Cl Na1Cl1 NaCl 2 2 Mg O 1 1 Mg O Mg1O1 MgO 2 1 Ca F 2 1 Ca F Ca1F2 CaF2 1 2 Li SO4 1 2 Li SO4 Li2(SO4)1 Li2SO4 3 3 Al PO4 1 1 Al PO4 Al1(PO4)1 AlPO4 Notes: a) You should be able to do all 5 steps in your head when you are finished the unit. b) When naming compounds, the least electronegative element is usually written first. c) Groups of elements such as SO42- and PO43- are referred to as radicals or polyatomic ions. These radicals behave as if they were a single entity and follow the cross over rule in the same manner as other single elements. Brackets are used in the formula only if there are 2 or more of the radical indicated in the formula. Binary Ionic Compounds A binary compound contains 2 elements only. Rules for Binary Ionic Compounds: a) The name of the binary compound always ends in "ide". b) The first mentioned element may have more than one valence and if it does this must be indicated in the name. c) Elements have a valence value determined by their group on the Periodic table unless indicated by the name. I 2 3 4 3 2 1 0 2 A) Binary Compounds where the first element has 1 valence only Use rule “c” and the cross-over rule. Examples: sodium oxide Na2O Exercise: calcium sulfide CaS magnesium bromide MgBr2 For all exercises you will give the name if formula provided and the formula if the name is provided. calcium nitride ________ silver sulphide ________ barium iodide ________ silicon oxide ________ zinc silicide ________ magnesium chloride ________ aluminum carbide ________ sodium fluoride ________ barium carbide ________ aluminum bromide ________ potassium bromide ________ cesium phosphide ________ LiCl _____________________________ K2S _____________________________ BaO _____________________________ Al2O3 _____________________________ B) Binary Compounds with Multivalent Elements Many transition metals and some other elements have multiple valence values and therefore can form multiple compounds with the same non-metal. Due to this it is necessary to indicate the valence of the metal in the name of the compound using either the Stock (IUPAC or Roman Numeral) or Classical (“ous” and “ic”) method. The second element (non-metal) always has a valence equal to the value as determined from its group in the periodic table. Method 1: Stock/IUPAC/Roman Numeral Method This is the currently preferred method as the valence of the first is indicated using the corresponding Roman Numeral in brackets following the name of the first element. Examples: iron(III) chloride FeCl3 tin(IV) iodide SnI4 phosphorus (III) oxide As2O3 mercury(II) oxide HgO Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. iron(III) chloride ___________ arsenic(V) iodide ___________ tin(IV) oxide ___________ gold(I) telluride ___________ phosphorus(V) chloride ___________ sulphur(VI) oxide ___________ copper(I) bromide ___________ bismuth(v) phosphide ___________ antimony(V) sulphide ___________ mercury(II) chloride ___________ arsenic(III) oxide ___________ gold(III) chloride ___________ mercury(I) sulphide ___________ copper(II)sulphide ___________ Pb3N2 ______________________________ SbF3 __________________________________ NiI2 ______________________________ MnO2 Co2Se3 ______________________________ BiF5 __________________________________ SnO2 ______________________________ ZnO _________________________________ _________________________________ 3 Method 2: Classical/"ous' and "ic" Method This method can be used when the multivalent element has 2 possible valences. The name of the element ending with "ous" denotes the lower valence value and the name of the element ending with an "ic" denotes the higher valence values. In some cases, the Latin name for the element is used. iron: gold: copper: tin: lead: cobalt nickel mercury platinum ferrous aurous cuprous stannous plumbous cobaltous nickelous mercurous platinous (valence = 2) (valence = 1) (valence = 1) (valence = 2) (valence = 2) (valence = 2) (valence = 2) (valence = 1) (valence = 2) and and and and and and and and and ferric auric cupric stannic plumbic cobaltic nickelic mercuric platinic (valence = 3) (valence = 3) (valence = 2) (valence = 4) (valence = 4) (valence = 3) (valence = 3) (valence = 2) (valence = 4) *Some elements having more than two valence values or oxidation states use specific values for the "ous" and the "ic". nitrogen manganese arsenic ous = 1 ic = 2 ous = 2 ic = 3 ous = 3 ic = 5 chromium phosphorus bismuth ous = 2 ic = 3 ous = 3 ic = 5 ous = 3 ic = 5 Examples: stannous chloride arsenic phosphide SnCl2 As3P5 phosphorous oxide nitric oxide P2O3 NO Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. ferric oxide _______________ auric iodide _______________ phosphoric sulfide _______________ nitrous phoshpide _______________ cuprous fluoride _______________ stannic oxide _______________ stannic fluoride _______________ arsenious nitride _______________ antimonous sulfide _______________ mercuric fluoride _______________ arsenious bromide _______________ cuprous nitride _______________ nickelic iodide _______________ auric chloride _______________ chromous oxide _______________ cupric sulfide _______________ NiI2 __________________________ Sb2S5 _________________________ HgBr __________________________ As4C5 _________________________ CoCl3 __________________________ Au2S _________________________ As3N5 __________________________ PAs _________________________ Fe2C __________________________ NiP _________________________ PbS __________________________ CoF3 _________________________ CuO __________________________ Au2Se3 _________________________ 4 Formula of Elements Most elements are written as single entities. Example: iron Fe(S) copper Cu(S) The exceptions to this rule are: a) the diatomic gases: H O F B I N Cl hydrogen H2(g) oxygen O2(g) nitrogen N2(g) chlorine Cl2(g) b) two other non-metals: helium He(g) fluorine F2(g) bromine Br2(l) sulphur S8(s) iodine I2(s) phosphorus P4(s) ** The exceptions must be memorized! ** Common Radicals or Polyatomic Ions Radicals or polyatomic ions are groups of atoms that are treated as a single entity when writing formulas and names. The following list of common radicals (polyatomic ions) must be memorized along with their valences. Positive polyatomic ions replace metals in an ionic compound and a negatively charged polyatomic ion goes in place of the nonmetal. If more than 1 of the radical is required in the chemical formula, brackets are placed around the radical’s formula with the subscript outside the bracket. Radical name Formula Valence Radical name Formula Valence 1 hydroxide OH 1 ammonium NH4+ cyanide 1 manganate MnO3- 1 chromate CNCrO22- 2 dichromate Cr2O72- 2 cyanate CNO- 1 thiocyanate SCN- 1 carbonate CO32- 2 nitrate NO3- 1 phosphate PO43- 3 sulfate SO42- 2 fluorate FO3- chlorate ClO3- 1 - Examples: sodium hydroxide lithium cyanide sodium dichromate NaOH LiCN Na2Cr2O7 1 potassium chromate ammonium hydroxide magnesium permanganate K2CrO4 NH4OH Mg(MnO4)2 Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. potassium hydroxide __________________ cobalt(II) cyanate __________________ iron(III) cyanate __________________ zinc hydroxide __________________ barium hydroxide __________________ gallium dichromate __________________ ammonium chloride __________________ potassium chromate __________________ copper(II) chromate __________________ Sn(CN)4 _______________________________ ammonium dichromate __________________ Al(OH)3 _______________________________ ammonium bromate __________________ KMnO4 _______________________________ ferrous hydroxide __________________ CsCN ________________________________ nickel(III) cyanide __________________ XeF6 ________________________________ auric bromide __________________ Ca(SCN)2 ______________________________ 5 Acids There are three groups of acids: - binary acids - oxy acids - derived oxy acids 1. Binary Acids This is a very small but common group of acids. The following rules apply to all of the members. 1. All have the prefix “hydro” and end with “ic”. 2. All must contain hydrogen as the first element. 3. Use the normal cross-over-rule to determine the formula. 4. All are dissociated in water and must be so indicated by using (aq) behind the formula. 5. Have no oxygen in their formula Examples: hydrochloric acid HCl(aq) hydrosulfuric acid H2S(aq) hydrocyanic acid HCN(aq) Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. hydrobromic acid ________ hydrotelluric acid ________ hydrofluoric acid ________ hydroiodic acid ________ hydroselenic acid ________ hydrosulfuric acid ________ hydrochloric acid ________ hydrophosphoric acid ________ 2. Oxy Acids This is a larger group of acids. They are alike in that they all contain H, O and at least one other non-metal element. All members of this group of acids follow the rules below. 1. The name of the acid ends in “ic”. 2. The name of the associated radical ends in “ate”. 3. The valence value of the associated radical is equal to the number of acidic hydrogens in the acid. The following 6 oxy acids and their associated radicals along with their valences must be memorized. Name of the Acid Formula of the Acid Name of the Formula of the valence value for the associated radical associated radical associated radical chromate C2H3O2- 1 1 acetic acid HC2H3O2 nitric acid HNO3 nitrate NO3- fluoric acid HFO3 fluorate FO3- 1 carbonic acid H2CO3 carbonate CO32- 2 2 3 sulfuric acid H2SO4 sulfate SO42- phosphoric acid H3PO4 phosphate PO43- NOTE: Using the periodic table it is possible to write the names and formulas for a number of other oxy acids using the fact that members of the same chemical family have similar chemical properties. Elements of the same chemical family (group) follow the pattern of the oxy acid immediately above the oxy acid that has been memorized. Example: memorized acid: fluoric acid = HFO3 therefore: chloric acid = HClO3 iodic acid = HIO3 bromic acid = HBrO3 If hydrogens are joined with the radicals learned earlier a few more acids can be formed. chromate = CrO42chromic acid = H2CrO4 cyanate = CNOcyanic acid = HCNO 6 Exercise: On a separate piece of paper complete the following chart for the acids listed. Name of the acid bromic acid manganic acid Formula of the acid telluric acid selenic acid Name of the Formula of the Valence value of the associated radical associated radical associated radical iodic acid cyanic acid silicic acid arsenic acid dichromic acid chromic acid 3. Derived Oxy Acids Theses acids are all derived from the parent oxy acids which you are to memorize. The table below shows the rule to determine the derived oxy acids from the parent acid. When adding or removing oxygen atoms from the parent acid, the number of hydrogens on the acid and the valence of the associated radical remains the same as the parent. Parent Oxy Acid + 1 O Parent Oxy Acid Parent Oxy Acid - 1 O Parent Oxy Acid - 2 O Type of Acid per______ic ic ous hypo_____ous Associated Radical per______ate ate ite hypo_____ite Example perchloric acid HClO4 perchlorate ion ClO4chloric acid HClO3 chlorate ion ClO3chlorous acid HClO2 chlorite ion ClO2hypochlorous acid HClO hypochlorite ion ClO- Exercise: On a separate piece of paper complete the following chart for the acids listed. Name of the acid Formula of the acid phosphoric acid phosphorous acid hypophosphorous acid sulfuric acid sulfurous acid nitric acid nitrous acid Name of the Formula of the Valence value of the associated radical associated radical associated radical perbromic acid bromic acid bromous acid hypobromous acid iodic acid iodous acid hypoiodous acid selenic acid selenous acid telluric acid tellurous acid manganic acid permanganic acid Salts The word, salt, is the general term given to a class of compounds which can be formed when an acid and a base neutralize each other. According to this definition then, most of the compounds we dealt with in the section on binary compounds could be considered to be salts formed from a base and a binary acid. In the same manner, we can get salts formed from a base and an oxy acid or derived oxy acid. Examples: sodium sulphate aluminum carbonate cupric nitrite Na2SO4 Al2(CO3)3 Cu(NO2)2 calcium phosphate zinc perchlorate stannous hypoiodite Ca3(PO4)2 Zn(ClO4)2 Sn(IO)2 7 Salts from oxy acids potassium chlorate____________________ sodium carbonate __________________ sodium nitrate ferric sulfate __________________ iron(III) acetate ____________________ gold(I) phosphate __________________ aluminum silicate ____________________ magnesium chlorate _________________________ magnesium phosphate __________________ ammonium nitrate _________________________ aurous sulfate K2CO3 _______________________________ ammonium chlorate ____________________ Na3PO4 _______________________________ zinc nitrate Fe(ClO3)2 _______________________________ potassium acetate _____________________ Al(C2H3O2)3 _______________________________ lithium chromate ______________________ Zn3(PO4)2 _______________________________ ____________________ ____________________ ________________________ Salts from Derived Oxy Acids aluminum sulfite __________________ copper(I) permanganate ____________________ cobalt(III) chlorite __________________ ammonium hypophosphite __________________ tin(IV) hypochlorite __________________ mercuric perchlorate _____________________ sodium phosphite __________________ magnesium sulfite _____________________ aluminum nitrite __________________ cuprous chlorite _____________________ cobalt(II) hypophosphite__________________ ammonium hypoiodite _____________________ stannic perchlorate __________________ gold(III) chromate sodium sulphite __________________ Ga(BrO4)3 ___________________________ aluminum chlorite __________________ Pt(ClO)4 ___________________________ nickel(III) hypochlorite __________________ Ag2O ___________________________ tin(IV) phosphite NH4CNO ___________________________ __________________ _____________________ Acid Radicals and Acid Salts 1. Acid Radicals In certain circumstances the oxy acids with multiple acidic hydrogens are able to lose the hydrogens one at a time. This results in radicals with acidic hydrogens still attached which are referred to as acid radicals. Step 1: H3PO4 H+ + H2PO4Step 2: H2PO4- H+ + HPO42- Step 3: HPO42- H+ + PO43Radical PO43HPO42H2PO4- Name phosphate monohydrogenphosphate dihydrogenphosphate Valence 3 2 1 Notes: a) Valence = # of hydrogen removed from the acid OR valence of non-acid radical-# of H’s still attached b) Mono may be left off a monohydrogen acid radical c) For oxy acids with 2 acidic hydrogens (diprotic acids), the prefix “bi” may be used in place of monohydrogen (eg. HCO3- = bicarbonate or monohydrogen carbonate) 8 For the following give the formula and valence value of the radicals listed: phosphite ___________ _____ monohydrogensulphate ___________ _____ monohydrogenphosphite ___________ _____ suphite ___________ _____ dihydrogenphosphite ___________ _____ monohydrogensulphite ___________ _____ hypophosphite ___________ _____ carbonate ___________ _____ monohydrogenhypophosphite ___________ _____ monohydrogencarbonate ___________ _____ dihydrogenhypophosphite ___________ _____ chromate ___________ _____ sulphate ___________ _____ monohydrogechromate ___________ _____ 2. Acid Salts The acid radicals are treated just like any other radical. Examples: calcium dihydrogenhypophosphite Ca(H2PO2)2 potassium monohydrogen carbonate KHCO3 aluminum bisulphite sodium hydrogensulfate Al(HSO3)3 NaHSO4 Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. sodium monohydrogenphosphate _____________ ferric monohydrogensulphate _______________ magnesium dihydrogenphosphate _____________ ammonium hydrogencarbonate ______________ aluminum dihydrogenphosphite _____________ chromium(III) hydrogensulphite ______________ chromium(III) hydrogenphosphite _____________ nickel(II) monohydrogensilicate ______________ cupric monohydrogenarsenate ______________ cadmium monohydrogenselenate ___________ stannic dihydrogenhypophosphite ____________ potassium monohydrogenselenite ___________ K2HPO3 ___________________________ Bi2(HPO3)5 __________________________ Ba(HSO3)2 Pb(H 2PO2)2 __________________________ ___________________________ Hydrates Hydrates are crystals containing a given number of water molecules within their structure. When naming a hydrate Greek prefixes in front of the word hydrate are used to indicate the number of water molecules into the crystal. In the chemical formula the number of water molecules is separated from the formula of the compound by a dot “∙” Examples: copper(II) sulfate pentahydrate CuSO4 ∙ 5H2O chlorine octahydrate Cl2 ∙ 8H2O Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. calcium sulfate dihydrate __________ chromium(III) nitrate nonahydrate __________ magnesium sulfite heptahydrate __________ cobalt(II) perchlorate pentahydrate __________ sodium carbonate decahydrate __________ barium chloride dihydrate __________ aluminum oxide monohydrate __________ aluminum nitrate monohydrate __________ ferric chloride hexahydrate __________ bromine decahydrate __________ calcium nitrate trihydrate __________ odine tetrahydrate __________ cadmium bromide tetrahydrate __________ copper(II) bromate hexahydrate __________ 9 Peroxides These are binary oxides, which contain an extra oxygen atom. Rules: 1. Write the formula of the regular oxide. 2. Add on one extra oxygen atom. 3. Do not at this stage cancel any of the subscripts. Na2O Na2O2 H2O H2O2 CaO CaO2 Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. zinc peroxide ____________________ potassium peroxide ____________________ calcium peroxide ____________________ strontium peroxide ____________________ cesium peroxide ____________________ hydrogen peroxide ____________________ copper(II) peroxide ____________________ barium peroxide ____________________ magnesium peroxide ____________________ aluminum peroxide ____________________ Thio Compounds The prefix “thio” in the name indicates that an oxygen atom has been replaced by a sulphur atom. Examples: potassium sulphate K2SO4 potassium thiosulphate K2S2O3 sodium carbonate Na2CO3 sodium thiocarbonate Na2SCO2 potassium cyanate KCNO potassium thiocyanate KSCN Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. ammonium thiocyanate _____________________ potassium thiosulfate ______________________ sodium monohydrogenthiosulphate ___________ aluminum thiocarbonate _____________________ CaS2O2 AlSPO3 ______________________________ _____________________________ Molecular Compounds These compounds contain 2 non-metals and are named using Greek prefixes. This method does not use the valence values. The Greek prefix is placed in front of the element name to indicate how many atoms of the element to place in the formula. If there is only 1 atom of the first element, mono is not used on the name of the first element. Prefixes 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca Examples: diphosphorus pentoxide P2O5 arsenic trichloride AsCl3 carbon monoxide CO xenon tetrafluoride XeF4 Exercise: For all exercises you will give the name if formula provided and the formula if the name is provided. xenon hexafluoride _________________ silicon dioxide _________________ sulfur dioxide _________________ carbon tetrachloride _________________ sulphur trioxide _________________ lead dioxide _________________ carbon dioxide _________________ manganese dioxide _________________ carbon disulphide _________________ nitrogen dioxide _________________ nitrogen tetroxide _________________ diphosphorus trisulfide _________________ 10 carbon tetrachloride _________________ ICl _______________________________ lead dioxide _________________ SeCl2 _______________________________ dichlorine monoxide _________________ ICl7 _______________________________ ___________ NF3 _______________________________ _______________________________ P2S5 _______________________________ diphosphorus pentasulphide KrF2 PRACTICE, PRACTICE, PRACTICE sulphur ___________________ lead (IV) nitride ___________________ plumbous peroxide ___________________ cupric chloride ___________________ calcium iodite ___________________ stannous iodide ___________________ sulfur dioxide ___________________ aluminum nitride ___________________ boron hypoarsenite ___________________ antimonous bromide ___________________ neon ___________________ tin (IV) sulfide ___________________ platinum (IV) thiocarbonite ___________________ silver oxide ___________________ calcium cyanide heptahydrate ___________________ calcium phosphide ___________________ cuprous permanganate ___________________ iron (II) sulfide ___________________ chromous acid ___________________ arsenic phosphide ___________________ cyanic acid ___________________ mercury (II) nitride ___________________ iodine monochloride ___________________ tin (IV) fluoride ___________________ stannic dihydrogen hypophosphite ________________ sodium hydride ___________________ auric peroxide ___________________ sodium monohydrogen phosphate _________________ ammonium selenide ___________________ barium hydroxide ___________________ titanium ___________________ sodium bicarbonate ___________________ ferrous thiosilicate ___________________ perchloric acid ___________________ cadmium bisulphate ___________________ nitrous acid ___________________ lithium phosphide ___________________ calcium peroxide ___________________ bromine ___________________ plumbous oxide ___________________ potassium oxide ___________________ potassium chloride ___________________ barium sulfide ___________________ hydrogen iodide ___________________ magnesium fluoride ___________________ antimonous phosphide ___________________ mercurous bromide ___________________ silver sulfide ___________________ zinc hydride ___________________ mercuric oxide ___________________ ferric sulfide ___________________ phosphorus (III) chloride ___________________ arsenic (V) oxide ___________________ ferrous oxide ___________________ 11 magnesium peroxide ___________________ silver hypoarsenite ___________________ zinc chromate dehydrate ___________________ copper (II) sulphate pentahydrate _________________ phosphoric acid ___________________ cyanic acid ___________________ chromic cyanide ___________________ sulfur dioxide ___________________ sodium thiosulfate hexahydrate ___________________ potassium permanganate ___________________ iron (III) carbonate ___________________ zinc chloride ___________________ magnesium phosphide ___________________ diarsenic pentasulfide ___________________ fluorine ___________________ hypophosphorous acid ___________________ chlorous acid ___________________ potassium hydroxide ___________________ dicarbon tetrahydride ___________________ iron (III) cyanate ___________________ hydrosulfuric acid ___________________ barium chromate ___________________ stannous tungstenate ___________________ ammonium thiocyanate ___________________ magnesium peroxide ___________________ potassium dichromate ___________________ ammonium hydrogenhypophosphite _______________ mercuric tellurite ___________________ tetracarbon decahydride ___________________ platinum (IV) silicate ___________________ nickelic perbromate ___________________ cobaltous thioselenite ___________________ chlorine dihydrate ___________________ tricarbon octahydride ___________________ copper (II) hypoarsenite ___________________ thiocyanic acid ___________________ dichromic acid ___________________ silver hypophosphite ___________________ aurous hydroxide ___________________ bromine ___________________ barium bithiocarbonate ___________________ silicon tetrahydride ___________________ hydrogen chloride ___________________ cesium arsenite dehydrate ___________________ sulfuric acid ___________________ manganic hydroxide ___________________ chromous cyanate ___________________ arsenious oxide ___________________ magnesium thiophosphate ___________________ silver chlorate ___________________ ferrous iodide ___________________ calcium perbromate ___________________ barium dichromate ___________________ barium phosphide ___________________ phosphorus ___________________ nickel (II) cyanide ___________________ periodic acid ___________________ beryllium phosphate ___________________ tetraphosphorus decaoxide ___________________ cadmium sulfite ___________________ hydrotelluric acid ___________________ boron tribromide ___________________ ammonium bitungstenate ___________________ silicon carbide ___________________ iron (III) peroxide ___________________ mercuric hydroxide ___________________ neon ___________________ cupric perchlorate ___________________ 12 gold (III) selenate ___________________ cupric acetate ___________________ stannic phosphide ___________________ acetic acid ___________________ arsenic (V) oxide ___________________ ammonium phosphate dehydrate _________________ sulfur trioxide ___________________ aurous chromate ___________________ hydrogen cyanide ___________________ ferrous hydroxide ___________________ lithium hypoiodite ___________________ stannic bromide ___________________ magnesium thiosilicate ___________________ plumbous thiocyanate ___________________ zinc phosphide ___________________ oxygen ___________________ aluminum peroxide ___________________ arsenic trihydride ___________________ lithium sulfate ___________________ ammonium dihydrogenphosphate _________________ magnesium hypochlorite ___________________ phosphorus pentachloride ___________________ manganese dioxide ___________________ silver nitrate ___________________ strontium nitrate ___________________ calcium chlorite ___________________ lead (IV) iodite ___________________ manganese (III) dichromate ___________________ zirconium chloride ___________________ potassium permanganate ___________________ cobalt (III) hypobromite ___________________ silicon dioxide ___________________ chromium (III) oxide ___________________ palladium (IV) nitrite ___________________ iodine monofluoride ___________________ diboron hexahydride ___________________ ammonium perbromate ___________________ sulfur ___________________ carbon disulphide ___________________ helium ___________________ aluminum hydroxide ___________________ uranium hexafluoride ___________________ beryllium sulfite ___________________ antimonic sulfate ___________________ lead (II) dihydrogenphosphite ___________________ phosphorus pentachloride ___________________ chromic hydrogenhypophosphite __________________ sulfur hexafluoride ___________________ iron (III) dihydrogenhypophosphite ________________ tin (II) cyanide ___________________ potassium hydrogenarsenite ___________________ phosphorus ___________________ mercury (II) bisulfate ___________________ chlorine ___________________ zinc monohydrogentellurite ___________________ cesium manganite ___________________ sodium bicarbonate ___________________ ammonium chromite ___________________ cobalt (III) bromite ___________________ calcium tungstenate ___________________ aluminum hydride ___________________ silver acetate hexahydrate ___________________ vanadium (V) oxide ___________________ potassium peroxide ___________________ carbon tetraiodide ___________________ sodium thiocarbonate ___________________ mercurous peroxide ___________________ lithium dithiosulfite ___________________ 13 PbS2O2 ________________________________ HIO4 (aq) ________________________________ MgO2 ________________________________ Ag2S ________________________________ Al2(SO2)3 ________________________________ Fe(OH)2 ________________________________ Au(MnO3)3 ________________________________ N2O4 ________________________________ BrCl5 ________________________________ K2S2O ________________________________ Ba(FO)2 ________________________________ HMnO4 (aq) ________________________________ ZnHAsO5 ________________________________ H2SiO3 (aq) ________________________________ Co(NO3)3 ________________________________ CaS2O2 ________________________________ HgSCO ________________________________ N2O4 ________________________________ H2S (aq) ________________________________ Au(IO4)3 ________________________________ SrCl2∙8 H2O ________________________________ PbO3 ________________________________ MnP ________________________________ Mg(MnO3)2 ________________________________ Cr(H2PO3)3 ________________________________ H2S (aq) ________________________________ K2O2 ________________________________ Fe(H2PO3)3 ________________________________ Al(ClO4)3 ________________________________ PCl5 ________________________________ Ga2(SiO3)3 ________________________________ Rb2SSiO2 ________________________________ P2S3 ________________________________ Ra3P2 · 7 H2O ________________________________ HNO3 (aq) ________________________________ NH4OH ________________________________ FeCr2O7 ________________________________ HCN (aq) ________________________________ CuSO4∙5H2O ________________________________ Ni3SPO2 ________________________________ H2SeO3 (aq) ________________________________ HTeO2 (aq) ________________________________ CaCr2O7 ________________________________ Cu2Se ________________________________ P2O5 ________________________________ Ga(OH)3 ________________________________ Cr(BrO)3 ________________________________ B2H6 ________________________________ MnO2 ________________________________ BaF2 ________________________________ Al2O4 ________________________________ H2CrO3 (aq) ________________________________ HI (aq) ________________________________ Pb(ClO)2 ________________________________ Au2(HAsO2)3 · 9 H2O _____________________________ H2Se(aq) ________________________________ SO3 ________________________________ CaMoO4 ________________________________ Li2SCO2 ________________________________ C4H8 ________________________________ Sr3N2 · 3 H2O ________________________________ ZnSeO2 ________________________________ (NH4)2SiO2 ________________________________ BaF2 ________________________________ H2Te (aq) ________________________________ H2Te(aq) ________________________________ Cr(CNO)3 ________________________________ Co2(HPO2)3∙4 H2O ______________________________ 14 NaBr ________________________________ SnCl4 __________________________________________________ Ca(C2H3O2)2 ________________________________ Cu2O __________________________________________________ P2O5 ________________________________ CuSO4 ________________________________ Ti(SO4)2 ________________________________ Ag2S ________________________________ FePO4 ________________________________ CsI ________________________________ K3N ________________________________ Al2S3 ________________________________ SO2 ________________________________ Ni(NO3)2 ________________________________ CuOH ________________________________ CoPO4 ________________________________ Zn(NO2)2 ________________________________ MnSO3 ________________________________ V2S3 ________________________________ Fe(ClO4)3 ________________________________ NaOH ________________________________ GaCl3 ________________________________ H2SO3 ________________________________ Cr2O3 ________________________________ H2S ________________________________ KNO2 ________________________________ H3PO4 ________________________________ Rb2CrO4 ________________________________ NH3 ________________________________ (NH4)2HPO4 ________________________________ HCN ________________________________ Cs2CO3 ________________________________ Ca(OH)2 ________________________________ Li2HPO4 ________________________________ Fe(OH)3 ________________________________ Mg(HCO3)2 ________________________________ H3P ________________________________ Ca(ClO3)2 ________________________________ Na2CO3 ________________________________ Sr(MnO4)2 ________________________________ P2O5 ________________________________ SCI3 ________________________________ NH3 ________________________________ Fe(C2H3O2)3 ________________________________ FeSO4 ________________________________ Ag2SO3 ________________________________ SiO2 ________________________________ Hg2Cl2 ________________________________ GaCl3 ________________________________ TiO2 ________________________________ CoBr2 ________________________________ (NH4)HSO4 ________________________________ B2H4 ________________________________ P2O5 ________________________________ CO ________________________________ NO2 ________________________________ P4 __________________________________________________ SO3 ________________________________ NH4Cl __________________________________________________ CCl4 ________________________________ Al2O3 __________________________________________________ Ag2Cr2O7 ________________________________ KBr __________________________________________________ CuSiO2 ________________________________ K2S __________________________________________________ AsBr5 ________________________________ SnI2 __________________________________________________ Sb3N5 ________________________________