Naming this molecule The Jeopardy champion! $200
advertisement

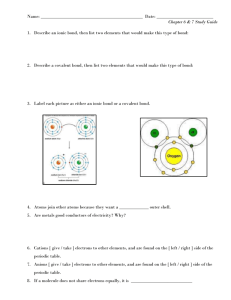
Atomic Structure Acids and Bases Periodic Table Let’s Bond Phases of Matter Take a Chance $200 $200 $200 $200 $200 $200 $400 $400 $400 $400 $400 $400 $600 $600 $600 $600 $600 $600 $800 $800 $800 $800 $800 $800 $1000 $1000 $1000 $1000 $1000 $1000 Final Jeopardy Category: Organic Chemistry Naming this molecule The Jeopardy champion! $200 24 C6H12O6 contains how many total atoms? $400 30.97376 What is the Atomic Mass of Phosphorus? $600 O Draw the Lewis Dot Diagram for Oxygen $800 Draw Bohr’s model of a Lithium atom 1e2e3P 4N $1000 Draw Bohr’s model of a Chlorine atom 7e8e2e17 P 18 N $200 Solution C What solution is the most acidic? Solution A has a pH of 13. Solution B has a pH of 4 Solution C has a pH of 3. Solution D has a pH of 14. $400 Base NaOH Acid or Base? $600 Tastes sour, pH less than 7, turns litmus red, reacts with most metals List 1 property of an acid $800 Acid + Base water + salt A neutralization reaction is an acid + ______ _____+ ______ $1000 H+ pH is a meaure of what ion? $200 3 (because C makes 4 total bonds) How many Hydrogens can go around each Carbon? C C $400 Halogens What is the name of the group 17 elements? $600 K, Potassium Name the element in the Alkali Group with 4 electron shells $800 (any of the Noble gases) – Xe, Ar, Kr, Rn Name one element that has physical properties similar to Ne $1000 H, N, O, F, Cl, Br, I Name the 7 diatomic elements $200 ionic What type of bond (ionic, covalent, or metallic) is shown below Na-Cl $400 covalent What type of bond (ionic, covalent, or metallic) is shown below C-C $600 Covalent - Carbon Tetrachloride Identify the type of bond and name the compound CCl4 $800 Covalent -- dinitrogen trioxide Identify the type of bond and name the compound N2 O 3 $1000 Ionic - Lithium Sulfate Identify the type of bond and name the compound Li2SO4 $200 physical Matter goes through Phase Changes. Are PHASE CHANGES chemical or physical? $400 Liquid Definite volume, indefinite shape describes a 1. Solid 2. Liquid 3. Gas $600 Nothing, the temp. will remain constant (B = melting and it’s a flat line Based on the picture below, if a cube of ice is melting and I add heat, what will happen to the temperature? $800 Solid and Liquid What phase(s) of matter are present at line B? $1000 1. Endo 2. exo 3. endo Endo or Exo 1. Solid Liquid 2. Gas Solid 3. Liquid Gas $200 B Changes that take place during a chemical reaction are shown in a A. chemical formula B. chemical equation C. chemical model D. chemical change $400 1 When a teaspoon sugar is added to water in a beaker, the sugar DISSOLVES, the resulting mixture is__________________. 1. a homogeneous solution 2. a compound 3. An element 4. a heterogeneous solution $600 1 2K2O → 4K + ______O 2 What number needs to go in front of O2 for this reaction to be balance? $800 Ca, Na, Sr Which of these elements will undergo a Single Replacement Reaction with HCl? Ca, Cu, Na, Sr, Au $1000 The mass are equal According to the law of conservation of mass, how does the mass (# of atoms) of the reactants compare with the mass (# of atoms) of the products in a chemical reaction?