Crizotinib outcomes in ALK- positive advanced NSCLC patients with brain metastases
Keith L. Davis, MA, 1 James A. Kaye, MD, DrPH, 2 Shrividya Iyer, PhD 2
1 RTI Health Solutions, Research Triangle Park, NC/United States of
America, 2 RTI Health Solutions, Waltham, MA/United States of
America, 3 Pfizer, Inc. New York, NY/United States of America
Mini Oral Presentation at the 16 th World Conference on Lung Cancer, Sep 6-9, 2015, Denver, CO, USA
Financial Disclosure: This study was sponsored by Pfizer, Inc.
Background
• Brain metastases are reported at initial diagnosis in 15-35% of patients with ALK + metastatic non-small cell lung cancer
(NSCLC) 1-3
• Frequency of brain lesions can increase (up to 46% of ALK + patients by one estimate 4 ) over the course of first-line therapy
• Crizotinib is an oral tyrosine kinase inhibitor (TKI) with proven efficacy against ALK + tumors 3,5
• Clinical benefits of crizotinib in ALK + metastatic NSCLC patients with brain metastases have been documented in trial data 6 and in single-case reports 7
1.
Doebele et al. Cancer 2012;118:4502-11. 2.
Kang et al. Respir Med 2014;108:388-94. 3. Shaw et al. N Engl J Med 2013;368:2385-94.
4. Weickhardt et al. J Thorac Oncol 2012;7:1807-1814. 5.
Ou et al. Ann Oncol 2014;25:415-22. 6.
Costa DB et al. J Clin Oncol 2015. [Epub ahead of print].
7.
Kinoshita Y et a.. BMJ Case Rep 2013. doi:10.1136/bcr-2013-200867.
2
3
Rationale and Objectives
• The clinical experience of crizotinib-treated patients with
ALK + metastatic NSCLC and brain metastases has not been widely assessed in real-world settings
• To help fill this research gap, we assessed the following in a small cohort of ALK + metastatic NSCLC patients with brain metastases:
– Demographic and clinical characteristics
– Objective response rate (ORR) of primary tumor during crizotinib treatment and 1-year survival rates from crizotinib initiation
– Status of brain lesions (intracranial response [ICR]) during crizotinib treatment
4
Methods
•
Retrospective chart review (anonymized data, IRB approved)
• Data abstraction performed in 2014 by pooled sample of 147 oncologists in the US (n = 107) and Canada (n = 40)
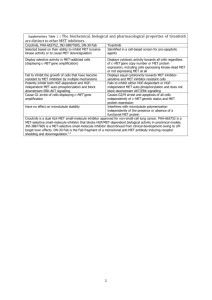
• Patient inclusion criteria:
–
Adults
(age ≥18) diagnosed with
ALK + metastatic NSCLC
–
Received crizotinib as first- or later-line treatment
–
First crizotinib treatment received between 8/1/2011 and 3/31/2013 (for US patients), or 4/12/2012 and
3/31/2013 (for Canadian patients)
–
Complete medical record through last crizotinib dose
–
Brain metastases present prior to or upon crizotinib initiation
Cohort for main study (n = 212)
See 2016 WCLC
Abstract No. 929
Cohort for present analyses (n = 33)
• Analyses were descriptive and exploratory
–
Kaplan-Meier (K-M) methods used for 1-year survival rate estimates
Results – Patient Demographics & Clinical Characteristics
5
Demographics
57.6 (11.9) Age at crizotinib initiation, mean (SD)
Sex, n (%)
Male
Female
19 (57.6)
14 (42.4)
Ethnicity, n (%)
White
Black
Asian/Pacific islander
25 (75.8)
5 (15.2)
3 (9.0)
Clinical Characteristics
Deceased at date of chart review, n (%)
10 (30.2)
Current/former smoker, n (%) 22 (66.7)
ECOG at diagnosis, n (%)
0 or 1
2 or 3
Adenocarcinoma histology, n
(%)
Crizotinib treatment duration
(days), median
18 (54.5)
15 (45.5)
28 (84.8)
230
Results – Best Response (Primary Tumor) and Survival
17%
18%
Patients Initiating Crizotinib as
First-Line Tx (n = 22)
4%
13%
48%
Complete response
Partial response
Stable disease
Disease progression
Not assessed
ORR = 61%
20%
Patients Initiating Crizotinib as
Second/Later-Line Tx (n = 11)
20%
60%
Complete response
Partial response
Stable disease
Disease progression
Not assessed
ORR = 60%
6
K-M Estimates of 1-Year Survival Probability from Crizotinib Initiation
Patients Initiating Crizotinib First-Line
Patients Initiating Crizotinib Second/Later-Line
0% 20% 40% 60%
80,7%
77,1%
80% 100%
Results – Status of Brain Lesions During Crizotinib Treatment
Intracranial Response During Crizotinib Treatment
100%
90%
80%
70%
60%
50%
40%
30%
20%
10%
0%
27%
14%
41%
18%
44%
11%
22%
22%
Stable Disease
Progressive Disease
Partial Intracranial
Response
Complete Intracranial
Response
First-Line Initiators (n = 22) Second-Line Initiators (n = 11)
• Note: 71% of patients received either whole brain radiotherapy or stereotactic radiosurgery prior to crizotinib initiation.
7
8
Study Limitations
•
Estimates based on small subsample (n = 33) of a larger multinational study
– Small sample size limited study to descriptive, exploratory analyses (no multivariable adjustments for covariates)
–
Results may not be generalizable to entire ALK + metastatic NSCLC population in the US or Canada
• No covariate adjustments
– ICR, for example, may be confounded by prior treatments (71% rec’d either whole brain radiotherapy or stereotactic radiosurgery)
• Convenience sample
– Non-randomized population
– Timing and manner of assessments of ORR and ICR not protocol-driven
(i.e., not assessed at pre-defined intervals, but rather at time points determined by the physicians in regular practice)
• Chart reviews subject to data entry/coding errors
Conclusions
• Complete or partial response of the primary tumor during crizotinib treatment was seen in a majority of patients (ORR
~60%)
• 1-year survival (~80%) was higher than a recent trial-based report of ALK + metastatic NSCLC patients with brain metastases (~65%) 6
• Results support emerging literature on the possible clinical benefits of crizotinib in ALK + metastatic NSCLC patients with brain metastases
• Findings provide signal that outcomes may be further optimized with earlier (first-line) initiation of crizotinib
6.
Costa DB et al. J Clin Oncol 2015. [Epub ahead of print].
9
 0
0